CRISPR in the Clinic: Unpacking the CTX310 ANGPTL3 Gene Editing Trial Design and Future Directions
This article provides a comprehensive analysis of the CTX310 clinical trial, a pioneering study investigating in vivo base editing of the ANGPTL3 gene for cardiovascular disease.
CRISPR in the Clinic: Unpacking the CTX310 ANGPTL3 Gene Editing Trial Design and Future Directions
Abstract
This article provides a comprehensive analysis of the CTX310 clinical trial, a pioneering study investigating in vivo base editing of the ANGPTL3 gene for cardiovascular disease. Targeted at researchers and drug development professionals, it explores the foundational science of ANGPTL3 as a therapeutic target, details the methodological blueprint of CTX310's trial design, examines critical troubleshooting and optimization challenges for gene editing therapies, and validates the approach through comparative analysis with other lipid-lowering strategies. The synthesis offers key insights into the translation of next-generation gene editing into viable clinical pathways.
ANGPTL3 as a Target: The Genetic and Mechanistic Rationale for CTX310's CRISPR-Based Approach
Rare human loss-of-function (LOF) mutations in the ANGPTL3 gene provide a validated genetic model for therapeutic targeting. Individuals carrying these mutations exhibit a sustained reduction in circulating low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and triglycerides (TGs) without apparent adverse sequelae. This "healthy depletion" phenotype establishes ANGPTL3 as a high-confidence target for reducing cardiovascular disease risk.
Table 1: Phenotypic Impact of ANGPTL3 Loss-of-Function in Human Carriers
| Parameter | Heterozygous Carriers (vs. Non-carriers) | Homozygous Carriers (vs. Non-carriers) | Key Studies |
|---|---|---|---|
| LDL-C | ↓ 17-27% | ↓ 60-70% | Stitziel et al., NEJM 2017; Musunuru et al., N Engl J Med 2020 |
| Triglycerides | ↓ 16-34% | ↓ 84% | Dewey et al., J Am Coll Cardiol 2017 |
| HDL-C | ↓ 4-12% | ↓ 62% | Minicocci et al., Eur Heart J 2013 |
| ASCVD Risk | ~40% reduction | Not established (rare) | Dewey et al., J Am Coll Cardiol 2017 |
This genetic evidence underpins the therapeutic hypothesis: pharmacological inhibition or genetic silencing of ANGPTL3 will recapitulate the beneficial cardiometabolic profile observed in LOF carriers.
Molecular Pathway and Mechanism
ANGPTL3 (Angiopoietin-like 3) is a secretory protein primarily synthesized in the liver. Its primary mechanism involves the inhibition of two key endothelial lipases: lipoprotein lipase (LPL) and endothelial lipase (EL). LPL is the principal enzyme responsible for hydrolyzing triglycerides in circulating chylomicrons and VLDL particles. EL primarily hydrolyzes phospholipids in HDL.
Diagram 1: ANGPTL3 Pathway and Lipase Inhibition (100 chars)
Inhibition of ANGPTL3 (via antibody, antisense oligonucleotide, or gene editing) disinhibits LPL and EL, leading to increased lipolysis of triglyceride-rich lipoproteins and altered HDL metabolism, resulting in the phenotype observed in LOF carriers.
Therapeutic Modulation & CTX310 Clinical Trial Context
CTX310 is an investigational CRISPR/Cas9-based in vivo gene-editing therapy designed to disrupt the ANGPTL3 gene in hepatocytes. The clinical trial design is based on translating the LOF genetics into a one-time therapeutic intervention.
Table 2: From Genetics to Therapy: Key Development Steps for CTX310
| Stage | Genetic Insight | Therapeutic Translation (CTX310) | Preclinical/Clinical Evidence |
|---|---|---|---|
| Target Validation | LOF carriers have low LDL-C, TG, HDL-C. | Disruption of ANGPTL3 gene in liver is hypothesized to mimic LOF. | NHP studies show >60% reduction in ANGPTL3 protein, with concomitant lipid lowering. |
| Dose Selection | Heterozygous vs. homozygous effect gradients. | Identify dose that achieves protein reduction >70% (akin to homozygote). | Phase I dose-escalation trial (NCTXXXXXXX) measuring ANGPTL3 levels and lipid panels. |
| Safety Monitoring | LOF carriers are generally healthy. | Monitor for theoretical risks: hepatic fat accumulation, platelet function. | Liver MRI-PDFF, standard safety labs, and coagulation panels in clinical trials. |
| Efficacy Endpoints | Lifelong reduction in lipids. | Primary: % change from baseline in fasting TG at 6 mo. Secondary: LDL-C, HDL-C, ANGPTL3 levels. | Interim data shows dose-dependent reductions up to 80% in TGs and 40% in LDL-C. |
Essential Research Protocols
Protocol 1: Quantification of ANGPTL3 Protein in Serum/Plasma
Purpose: To measure therapeutic knockdown of ANGPTL3 following intervention (e.g., CTX310). Method: ELISA. Detailed Steps:
- Sample Prep: Collect human or NHP serum/plasma in EDTA tubes. Centrifuge at 1500 x g for 15 min at 4°C. Aliquot and store at -80°C. Avoid repeated freeze-thaw cycles.
- ELISA Procedure: Use a validated commercial human ANGPTL3 ELISA kit (e.g., R&D Systems DY7825). a. Bring all reagents and samples to room temperature. b. Add 100 µL of Assay Diluent to each well of the pre-coated plate. c. Add 50 µL of standard or sample per well. Cover and incubate 2 hours at RT on a horizontal shaker. d. Aspirate and wash each well 4 times with Wash Buffer. e. Add 200 µL of ANGPTL3 Conjugate per well. Incubate 2 hours at RT. f. Aspirate and wash 4 times. g. Add 200 µL of Substrate Solution. Incubate 30 minutes at RT, protected from light. h. Add 50 µL of Stop Solution. Read absorbance at 450 nm with correction at 540 or 570 nm within 30 minutes.
- Analysis: Generate a standard curve using 4-parameter logistic (4-PL) curve fit. Report concentrations in ng/mL.
Protocol 2: In Vitro Assessment of LPL Activity Modulation
Purpose: To functionally confirm the biological consequence of ANGPTL3 suppression in conditioned media. Method: Fluorescent LPL Activity Assay. Detailed Steps:
- Cell Culture & Conditioned Media: Seed HepG2 cells in a 6-well plate. Transfert with control or ANGPTL3-targeting editors (e.g., CRISPR RNPs). After 72h, collect serum-free conditioned media. Centrifuge to remove debris.
- LPL Activity Reaction: a. Prepare a working solution of fluorescent triglyceride substrate (e.g., EnzChek Lipase Substrate, Invitrogen E33955). b. In a black 96-well plate, mix 50 µL of conditioned media with 50 µL of substrate working solution. c. Include controls: Blank (substrate + buffer), Positive Control (substrate + purified LPL enzyme). d. Cover plate and incubate at 37°C for 30-60 minutes, protected from light.
- Measurement: Read fluorescence (ex/em ~485/535 nm) on a plate reader. Activity in test samples is proportional to fluorescence intensity, indicating LPL disinhibition due to reduced ANGPTL3 in conditioned media.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Key Reagents for ANGPTL3 Pathway and Editing Research
| Reagent/Material | Supplier Examples | Function in Research |
|---|---|---|
| Recombinant Human ANGPTL3 Protein | R&D Systems, Sino Biological | Positive control for assays; for in vitro inhibition studies. |
| Anti-ANGPTL3 Antibodies (ELISA/Neutralizing) | R&D Systems, MilliporeSigma | Quantification (ELISA) or functional blockade in mechanistic studies. |
| Human ANGPTL3 ELISA Kit | R&D Systems, Abcam | Gold-standard for measuring ANGPTL3 protein levels in biological fluids. |
| Lipoprotein Lipase (LPL) Activity Assay Kit | Invitrogen, Cell Biolabs | Functional readout of pathway modulation via fluorescent or colorimetric methods. |
| CRISPR/Cas9 Reagents (sgRNAs, Cas9 Nuclease) | Synthego, IDT, Thermo Fisher | For creating ANGPTL3 knockout cell lines to model LOF in vitro. |
| Primary Human Hepatocytes | Lonza, BioIVT | Physiologically relevant in vitro model for liver-targeted editing studies. |
| Next-Gen Sequencing Kit for INDEL Analysis | Illumina, Thermo Fisher | Measuring editing efficiency at the ANGPTL3 locus in genomic DNA. |
Diagram 2: CTX310 Clinical Development Logic (95 chars)
The CTX310 clinical trial represents a pivotal investigation into a next-generation lipid management strategy via in vivo base editing of ANGPTL3. This approach aims to induce a sustained, loss-of-function phenotype, mimicking the cardioprotective effects observed in human genetic carriers of ANGPTL3 deficiency. Within the broader thesis on CTX310 trial design, this application note details the preclinical and translational research protocols essential for validating the mechanistic rationale and therapeutic potential of ANGPTL3 editing, moving beyond the established paradigm of PCSK9 inhibition.
Table 1: Comparative Lipid-Lowering Efficacy of PCSK9 vs. ANGPTL3 Targeting Strategies
| Parameter | PCSK9 Monoclonal Antibody | PCSK9 siRNA (Inclisiran) | ANGPTL3 mAb (Evinacumab) | ANGPTL3 Editing (Preclinical/CTX310 Goal) |
|---|---|---|---|---|
| Primary Target | Extracellular PCSK9 | Hepatic PCSK9 mRNA | Extracellular ANGPTL3 Protein | Hepatic ANGPTL3 Gene |
| LDL-C Reduction | ~60% | ~50% | ~50% (in HoFH) | >50% (sustained) |
| Triglyceride Reduction | Minimal (~10%) | Minimal | ~50% | >50% (sustained) |
| Dosing Frequency | Every 2-4 weeks | Twice yearly | Monthly | Single dose, potentially durable |
| Mechanism | Protein blocking | RNA interference | Protein blocking | Precise genomic editing |
Table 2: Key Biomarkers in ANGPTL3-Loss-of-Function Research
| Biomarker | Function | Change in ANGPTL3 Inhibition/Editing | Clinical Significance |
|---|---|---|---|
| ANGPTL3 (serum) | LPL & EL inhibitor | Drastically reduced | Primary pharmacodynamic readout |
| LDL-C | Atherogenic cholesterol | Reduced 40-50% | Primary efficacy endpoint |
| Triglycerides | Circulating fat particles | Reduced 40-60% | Key efficacy endpoint |
| HDL-C | "Good" cholesterol | Mild increase | Potential favorable effect |
| Lipoprotein(a) | Independent risk factor | Potentially reduced | Differentiating benefit vs. PCSK9i |
| VLDL & Remnants | Atherogenic remnants | Reduced | Addresses residual cardiovascular risk |
Detailed Application Notes & Protocols
Protocol 1: In Vitro Validation of ANGPTL3 Base Editor Activity
Objective: To assess the efficiency and precision of CTX310-like base editors in human hepatocyte cell lines. Workflow:
- Cell Culture: Maintain HepG2 or primary human hepatocytes in appropriate media.
- Editor Delivery: Transfect cells with lipid nanoparticles (LNPs) encapsulating base editor mRNA and sgRNA targeting the human ANGPTL3 splice site or introducing a premature stop codon.
- Harvest: Collect cells and media at 72h and 7 days post-transfection.
- Genomic Analysis:
- Extract genomic DNA.
- Perform targeted deep sequencing (>10,000x coverage) of the ANGPTL3 locus to quantify editing efficiency and assess for potential off-target editing using pre-defined computational predictions (e.g., Cas-OFFinder).
- Functional Readout:
- Extract total RNA for qRT-PCR to measure ANGPTL3 mRNA knockdown.
- Analyze cell culture supernatant via ELISA to quantify ANGPTL3 protein secretion reduction. Expected Outcome: >80% allelic editing with >99% precision, leading to >90% reduction in ANGPTL3 protein secretion.
Protocol 2: In Vivo Efficacy & Durability Study in Humanized Mouse Models
Objective: To evaluate the pharmacokinetics, pharmacodynamics, and durability of a single LNP dose of ANGPTL3 base editor. Workflow:
- Animal Model: Use a humanized mouse model expressing the human ANGPTL3 gene (e.g., Angptl3-/- mice with AAV-mediated human ANGPTL3 liver expression).
- Dosing: Administer a single intravenous dose of LNP-formulated base editor (dose range: 1-3 mg/kg mRNA equivalent). Include vehicle and non-targeting sgRNA controls.
- Longitudinal Monitoring:
- Weekly/Biweekly: Collect tail vein blood.
- Serum Analysis: Measure human ANGPTL3 protein (ELISA), full lipid panel (LDL-C, TG, HDL-C), and liver enzymes (ALT/AST).
- Terminal Analysis (e.g., Week 12 & 24):
- Isolate liver tissue for genomic DNA analysis (editing efficiency, deep sequencing).
- Isolate hepatocytes for RNA-seq to confirm on-target and assess transcriptomic changes. Expected Outcome: Single dose leads to >70% reduction in serum ANGPTL3 and lipids, sustained for >6 months, with normal liver transaminases.
Protocol 3: Assessment of ANGPTL3 Editing on Lipoprotein Metabolism
Objective: To mechanistically confirm that ANGPTL3 editing enhances lipoprotein lipase (LPL) and endothelial lipase (EL) activity. Workflow:
- Post-Heparin Plasma Collection: From Protocol 2 animals, collect plasma pre- and post-intravenous heparin injection (to release vascular LPL/EL).
- Lipase Activity Assays:
- LPL Activity: Use a fluorogenic TG substrate assay in the presence of a specific LPL inhibitor to confirm specificity.
- EL Activity: Use a radiolabeled phospholipid substrate assay.
- Fast Protein Liquid Chromatography (FPLC): Perform lipoprotein fractionation of pooled mouse plasma to visualize reductions in VLDL and LDL particle peaks. Expected Outcome: Significant increase in post-heparin LPL and EL activity in treated animals, correlating with reduced VLDL and LDL fractions on FPLC.
Diagrams
Diagram Title: ANGPTL3 Pathway and Editing Mechanism
Diagram Title: Preclinical In Vivo Study Protocol
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for ANGPTL3 Editing Research
| Reagent/Material | Provider Examples | Function in Protocol |
|---|---|---|
| Human ANGPTL3 ELISA Kit | R&D Systems, Abcam | Quantifies ANGPTL3 protein reduction in serum/cell media (Protocols 1 & 2). |
| Lipid Profile Assay Kit | Wako Diagnostics, Sigma-Aldrich | Measures LDL-C, TG, HDL-C in mouse/human plasma (Protocol 2). |
| LPL Activity Assay Kit | Cell Biolabs, Roar Biomedical | Fluorometrically quantifies LPL activity in post-heparin plasma (Protocol 3). |
| Fast Protein Liquid Chromatography (FPLC) System | Cytiva (ÄKTA) | Separates lipoprotein particles (VLDL, LDL, HDL) by size (Protocol 3). |
| Next-Gen Sequencing Library Prep Kit | Illumina, IDT | Prepares amplicons for deep sequencing of target & off-target sites (Protocol 1). |
| LNP Formulation Reagents | Precision NanoSystems | For encapsulating base editor mRNA/sgRNA complexes for in vivo delivery (Protocol 2). |
| Humanized ANGPTL3 Mouse Model | Jackson Laboratory, Cyagen | In vivo model expressing human ANGPTL3 for translational studies (Protocol 2). |
| Primary Human Hepatocytes | Lonza, Thermo Fisher | Physiologically relevant in vitro model for editing validation (Protocol 1). |
The clinical development of CTX310, an investigational in vivo base editing therapy for targeting the ANGPTL3 gene, represents a pivotal application of evolved genome editing technologies. The broader thesis within its trial design posits that precision base editors, by enabling efficient, predictable, and potentially safer correction of disease-driving variants (e.g., ANGPTL3 loss-of-function for cardiovascular disease), offer a superior therapeutic profile compared to earlier CRISPR-Cas9 nuclease approaches. This protocol details the critical in vitro and preclinical experiments that validate the transition from conventional CRISPR-Cas9 to adenine base editors (ABEs) for ANGPTL3 targeting, underpinning the rationale for CTX310.
The following table compares key performance metrics of SpCas9 nuclease versus an ABE (e.g., ABE8e) for introducing a precise A•T to G•C edit at the target ANGPTL3 locus in human hepatocyte cell lines (e.g., HepG2).
Table 1: Comparison of Editing Outcomes for ANGPTL3 Modification
| Parameter | CRISPR-Cas9 Nuclease | Adenine Base Editor (ABE8e) |
|---|---|---|
| Primary Edit Rate | Indel frequency: 60-80% | Target A-to-G conversion: 50-70% |
| Precise Desired Edit | <5% (via HDR, requires donor) | 50-70% (direct, no donor required) |
| Undesired Indel Byproducts | 60-80% (primary product) | Typically <3% |
| Transversion/Other Base Subs | Rare | <1.5% (mostly bystander edits) |
| Therapeutic Product Purity | Low | High |
Detailed Experimental Protocols
Protocol 1: Design and Validation of gRNAs for ANGPTL3 Base Editing Objective: Identify optimal spacer sequences for ABE targeting of the human ANGPTL3 gene to create a loss-of-function variant. Materials: See "The Scientist's Toolkit" below. Procedure:
- Target Identification: Align human and relevant preclinical model ANGPTL3 genomic sequences. Identify target adenines within the canonical NGG PAM for SpCas9-derived ABE, prioritizing early exonic regions to introduce nonsense or splice-disrupting codons.
- gRNA Cloning: Synthesize oligos for candidate spacer sequences (20-nt). Anneal and ligate into the BsmBI site of a plasmid co-expressing the ABE8e protein and the sgRNA (e.g., pCMV_ABE8e-sgRNA).
- Cell Transfection: Seed HepG2 cells in a 24-well plate. At 70-80% confluency, transfect with 500 ng of each ABE8e-sgRNA plasmid using a lipid-based transfection reagent. Include a non-targeting sgRNA control.
- Genomic DNA Harvest: 72 hours post-transfection, extract genomic DNA using a silica-membrane-based kit.
- PCR Amplification: Amplify the target region (~300-500 bp) using high-fidelity PCR.
- Next-Generation Sequencing (NGS) Analysis: Purify PCR products, prepare NGS libraries, and sequence on a MiSeq system. Analyze data using a base editing analysis tool (e.g., BEAT) to calculate:
- A-to-G editing efficiency at the target position.
- Bystander edit rates at other adenines within the editing window.
- Indel frequency.
Protocol 2: Assessment of ANGPTL3 Protein Knockdown and Functional Phenotype Objective: Confirm that ABE-mediated editing leads to reduced ANGPTL3 protein and the intended functional effect (e.g., reduced extracellular lipoprotein lipase inhibition). Procedure:
- Generate Edited Cell Pool: Scale up transfection of the lead ABE8e-sgRNA construct in HepG2 cells. Culture for 7 days to allow protein turnover.
- Western Blot Analysis:
- Lyse cells in RIPA buffer.
- Resolve 20 µg of total protein on a 4-12% Bis-Tris gel and transfer to PVDF membrane.
- Probe with anti-ANGPTL3 and anti-β-actin primary antibodies.
- Quantify band intensity; expect >80% reduction in ANGPTL3 protein for a high-efficiency editor.
- Functional Lipoprotein Lipase (LPL) Activity Assay:
- Collect conditioned media from edited and control cells.
- Incubate media with recombinant LPL and a fluorescent triglyceride substrate.
- Measure liberated fluorescence over time. Edited cell media should show increased LPL activity due to loss of ANGPTL3-mediated inhibition.
Pathway and Workflow Visualizations
Diagram Title: ANGPTL3 Editing Therapeutic Pathway
Diagram Title: gRNA Screening and Validation Protocol
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for ANGPTL3 Base Editing Research
| Reagent/Material | Function & Rationale | Example Vendor/ID |
|---|---|---|
| ABE Expression Plasmid | All-in-one vector for delivery of base editor (e.g., ABE8e) and sgRNA. Critical for efficient co-expression. | Addgene #138489 (pCMV_ABE8e) |
| Human Hepatocyte Cell Line | In vitro model for human liver biology and ANGPTL3 expression/secret ion. | HepG2 or HepaRG cells |
| Lipid-Based Transfection Reagent | High-efficiency delivery of plasmid DNA to hepatoma cells. | Lipofectamine 3000 |
| High-Fidelity PCR Polymerase | Accurate amplification of target genomic locus for NGS analysis without introducing errors. | Q5 Hot-Start Polymerase |
| NGS Library Prep Kit for Amplicons | Prepares purified PCR amplicons for high-throughput sequencing to quantify editing. | Illumina DNA Prep Kit |
| Anti-ANGPTL3 Antibody | Validated primary antibody for detection of ANGPTL3 protein knockdown via Western blot. | Rabbit monoclonal, CST # |
| Lipoprotein Lipase Activity Assay Kit | Functional biochemical assay to measure the consequence of ANGPTL3 loss on LPL inhibition. | Fluorometric LPL Activity Kit |
Application Notes: In Vivo Efficacy and Mechanism of Action
The clinical development of CTX310, a CRISPR/Cas-based gene-editing therapeutic for durable ANGPTL3 knockdown, is predicated on robust preclinical evidence from animal models. These studies establish a clear mechanistic link between ANGPTL3 inhibition, profound reduction of atherogenic lipoproteins, and protection from atherosclerosis. The data validate ANGPTL3 as a high-value target for a single-course treatment paradigm.
Key Findings:
- Lipid-Lowering Efficacy: Genetic deletion or antisense oligonucleotide (ASO)-mediated knockdown of ANGPTL3 in mice results in reductions of plasma triglycerides (TG) by 30-60% and low-density lipoprotein cholesterol (LDL-C) by 20-50%, depending on the model and method.
- Atheroprotection: In hyperlipidemic models (e.g., Ldlr-/- or Apoe-/- mice), ANGPTL3 deficiency or knockdown significantly reduces aortic root atherosclerotic lesion area by 30-70% compared to controls, with improvements in plaque stability markers.
- Mechanistic Insights: ANGPTL3 inhibition primarily increases lipoprotein lipase (LPL) and endothelial lipase (EL) activity, enhancing the catabolism of TG-rich lipoproteins (TRLs) and subsequent receptor-mediated clearance of remnants. This multi-lipoprotein targeting is a distinct advantage over statins.
Implications for CTX310 Trial Design: These data support the investigation of CTX310 in patients with refractory hypercholesterolemia or familial hypercholesterolemia (FH), predicting a durable, pan-lipid-lowering effect that translates to reduced cardiovascular risk. Dose-ranging studies can be informed by the graded responses observed in knockdown models.
Table 1: Lipid-Lowering Effects of ANGPTL3 Modulation in Mouse Models
| Model / Intervention | Species/Strain | TG Reduction (%) | LDL-C Reduction (%) | HDL-C Change (%) | Primary Reference |
|---|---|---|---|---|---|
| Angptl3-/- (full knockout) | C57BL/6J | ~50% | ~35% | ±10% (ns) | Koishi et al., 2002 |
| Angptl3-/- on HFD | C57BL/6J | ~60% | ~30% | +15% | Wang et al., 2015 |
| Ldlr-/- + Angptl3 ASO | C57BL/6J background | ~55% | ~50% | -20% | Graham et al., 2017 |
| Apoe-/- + Angptl3 siRNA | C57BL/6J background | ~40% | ~25% | ±5% (ns) | Zhang et al., 2022 |
| Ldlr-/-; Angptl3-/- (DKO) | C57BL/6J background | ~75% | ~70% | -25% | Gusarova et al., 2018 |
Table 2: Atheroprotective Outcomes in Hyperlipidemic Models
| Model / Intervention | Atherosclerosis Metric | Reduction vs. Control | Plaque Stability Indicators |
|---|---|---|---|
| Ldlr-/- + Angptl3 ASO | Aortic Root Lesion Area (Oil Red O) | ~65% | ↑ Collagen; ↓ Macrophage area |
| Apoe-/-; Angptl3-/- (DKO) | En Face Lesion Area (Whole Aorta) | ~50% | ↑ Fibrous cap; ↓ Necrotic core |
| Ldlr-/- + Anti-ANGPTL3 mAb | Aortic Arch Lesion Area (H&E) | ~35% | ↑ Smooth muscle cell content |
| Apoe-/- + Angptl3 siRNA | Aortic Root Cross-Sectional Area | ~30% | ↓ Intraplaque hemorrhage |
Detailed Experimental Protocols
Protocol 1: Evaluating ANGPTL3 Knockdown Efficacy with GalNAc-Conjugated siRNA in a Hyperlipidemic Mouse Model
Objective: To assess the potency and durability of hepatocyte-targeted ANGPTL3 knockdown on plasma lipids. Materials: Ldlr-/- mice (8-week-old, male), ANGPTL3-targeting GalNAc-siRNA (or control), isoflurane, heparinized capillary tubes, automated clinical chemistry analyzer. Procedure: 1. House mice on a high-fat diet (HFD, 1.25% cholesterol) for 2 weeks prior to intervention. 2. Randomize mice into treatment (n=10) and control (n=10) groups. Baseline blood samples are collected via retro-orbital bleed under anesthesia. 3. Administer a single subcutaneous injection of ANGPTL3 GalNAc-siRNA (e.g., 3 mg/kg) or PBS control. 4. Collect serial blood samples at weeks 1, 2, 4, 8, and 12 post-injection after a 4-hour fast. 5. Isolate plasma by centrifugation. Quantify TG, total cholesterol, LDL-C, and HDL-C using enzymatic assays on a clinical analyzer. 6. Terminally, harvest liver tissue. Isolate RNA and quantify Angptl3 mRNA levels via qRT-PCR for confirmation of target engagement. Analysis: Compare longitudinal lipid profiles and terminal mRNA levels between groups using two-way ANOVA.
Protocol 2: Histopathological Assessment of Atherosclerotic Lesions
Objective: To quantify the atheroprotective effect of ANGPTL3 knockdown. Materials: Optimal Cutting Temperature (OCT) compound, cryostat, Oil Red O stain, hematoxylin, fluorescently-labeled antibodies (anti-MOMA2 for macrophages, anti-alpha-SMA for smooth muscle cells), confocal microscope. Procedure: 1. Following Protocol 1 (12-week endpoint), perfuse mice with PBS, then excise the heart and ascending aorta. 2. Embed the aortic root in OCT compound. Serially section (10 µm thickness) through the aortic valve leaflets. 3. Stain sections with Oil Red O (lipid) and Hematoxylin (nuclei). Image 5-8 sections per mouse. 4. Quantify total lesion area per section using image analysis software (e.g., ImageJ). 5. For immunofluorescence, stain adjacent sections with MOMA2 and alpha-SMA antibodies. Quantify positive areas. Analysis: Report mean lesion area per group and the ratio of alpha-SMA+ to MOMA2+ area as a stability index. Use unpaired t-tests for comparison.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for ANGPTL3 Preclinical Research
| Reagent / Material | Function / Application | Example Vendor |
|---|---|---|
| ANGPTL3 KO/KI Mice | Genetically engineered models for loss-of-function or humanized studies. | Jackson Laboratory |
| GalNAc-conjugated ANGPTL3 siRNA | Liver-targeted, durable knockdown tool for mimicking therapeutic effect. | Dicerna, Alnylam |
| Recombinant ANGPTL3 Protein | For in vitro rescue experiments, LPL/EL activity inhibition assays. | R&D Systems |
| Anti-ANGPTL3 Monoclonal Antibody | For immunoblotting, ELISA, and functional blocking studies. | Thermo Fisher |
| LPL Activity Assay Kit | Fluorometric or colorimetric kit to measure LPL activity in post-heparin plasma or tissue. | Cell Biolabs |
| Fast Protein Liquid Chromatography (FPLC) System | For detailed lipoprotein profile analysis (VLDL, LDL, HDL separation). | GE Healthcare |
Pathway and Workflow Diagrams
Diagram 1: ANGPTL3 Inhibits Lipoprotein Clearance
Diagram 2: Preclinical Workflow for CTX310
Blueprint for Editing: A Deep Dive into the CTX310 Clinical Trial Design and Protocol
Application Notes: CTX310 Clinical Trial Design for ANGPTL3 Editing
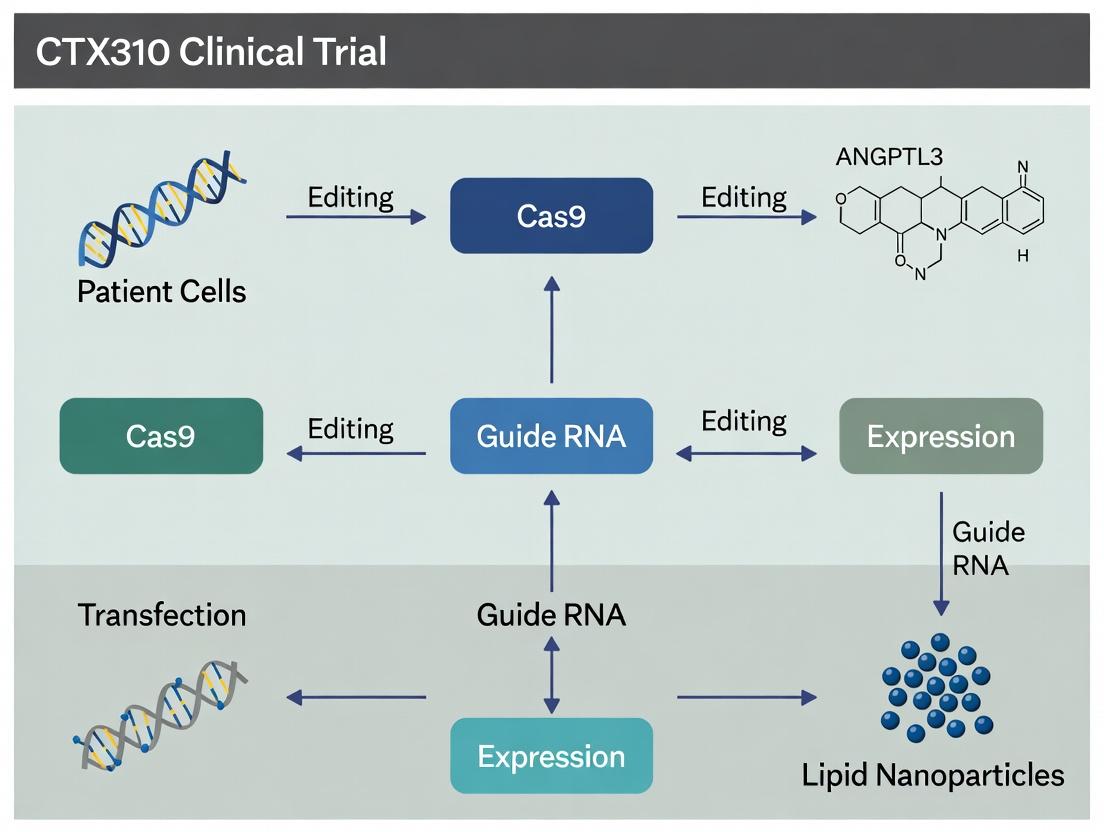
This document outlines the integrated Phase I/II trial design for CTX310, an investigational CRISPR-based in vivo gene-editing therapeutic targeting ANGPTL3 for the treatment of homozygous familial hypercholesterolemia (HoFH) and severe hypertriglyceridemia.
Rationale for an Integrated Phase I/II Design
An integrated design accelerates development by combining safety, tolerability, pharmacokinetics/pharmacodynamics (PK/PD), and preliminary efficacy assessments. For a first-in-human gene editor, this allows for dose-finding (Phase I) and initial signal detection (Phase II) within a single protocol framework, optimizing resource use and patient exposure.
Trial Design Schema
The study is a multi-center, open-label, dose-escalation and cohort-expansion study.
Table 1: Trial Structure Overview
| Component | Description |
|---|---|
| Phase | Integrated Phase I/II (First-in-Human) |
| Design | Open-label, Sequential Cohort, Dose Escalation with Expansion |
| Study Drug | CTX310 (CRISPR-Cas9 lipid nanoparticle formulation targeting ANGPTL3 in hepatocytes) |
| Population | Adults (18-75) with HoFH or severe hypertriglyceridemia (≥500 mg/dL) despite standard care |
| Parts | Part A: Single Ascending Dose (SAD) Escalation (4 planned cohorts). Part B: Cohort Expansion at Selected Dose(s). |
Table 2: Planned Dose Cohorts (Part A: SAD)
| Cohort | Number of Participants | CTX310 Dose Level | Primary Objectives |
|---|---|---|---|
| 1 | 3-4 | 0.1 mg/kg | Safety, Tolerability, PK |
| 2 | 3-4 | 0.3 mg/kg | Safety, Tolerability, PK, PD (target editing) |
| 3 | 3-4 | 1.0 mg/kg | Safety, Tolerability, PK, PD, Preliminary Efficacy |
| 4 | 3-4 | 3.0 mg/kg | Safety, Tolerability, PK, PD, Preliminary Efficacy |
Primary & Secondary Endpoints
Table 3: Primary and Key Secondary Endpoints
| Endpoint Category | Specific Endpoint | Measurement Timepoint |
|---|---|---|
| Primary (Safety) | Incidence and severity of Treatment-Emergent Adverse Events (TEAEs) and Serious Adverse Events (SAEs) | Day 1 through Week 52 |
| Secondary (PK/PD) | Plasma concentration of CTX310 components (gRNA, LNP lipids) | Pre-dose to 48 hours |
| Secondary (Activity) | ANGPTL3 gene editing rate in circulating protein-associated DNA | Weeks 4, 12, 24, 52 |
| Secondary (Efficacy) | Percent change from baseline in fasting: 1) Triglycerides (TG), 2) LDL-Cholesterol (LDL-C), 3) ANGPTL3 protein level | Weeks 4, 12, 24, 52 |
Detailed Experimental Protocols
Protocol 4.1: Assessment ofANGPTL3Editing (Primary PD Endpoint)
Objective: Quantify the frequency of insertions/deletions (indels) at the ANGPTL3 target locus in DNA isolated from plasma. Methodology:
- Sample Collection: Collect whole blood in EDTA tubes at screening (baseline) and post-treatment timepoints. Centrifuge at 1600×g for 10 min to isolate plasma.
- Cell-Free DNA (cfDNA) Isolation: Use the QIAamp Circulating Nucleic Acid Kit (Qiagen). Add 5 mL plasma to lysis buffer, bind to silica membrane, wash, and elute in 50 µL Buffer AVE.
- Target Amplification: Perform nested PCR using primers flanking the CTX310 cut site in ANGPTL3 exon 1. Use high-fidelity polymerase (KAPA HiFi).
- Next-Generation Sequencing (NGS) Library Prep: Barcode amplified products from individual timepoints. Purify using AMPure XP beads.
- Sequencing & Analysis: Pool libraries and sequence on Illumina MiSeq (2x150 bp). Align reads to human genome (hg38). Use CRISPResso2 pipeline to quantify % indels within a 10 bp window of the predicted cut site. Editing rate = (indel reads / total aligned reads) × 100%.
Protocol 4.2: Efficacy Biomarker Analysis (Triglycerides & ANGPTL3 Protein)
Objective: Measure changes in serum triglycerides and circulating ANGPTL3 protein. Methodology (Triglycerides):
- Perform a 12-hour fast prior to blood draw.
- Collect serum in clot activator tubes. Allow to clot for 30 min, centrifuge at 1300×g for 10 min.
- Analyze using enzymatic colorimetric assay (Roche Cobas c502) following manufacturer's protocol. Methodology (ANGPTL3 Protein):
- Use serum from step 2 above.
- Employ a validated quantitative sandwich ELISA (Human ANGPTL3 Quantikine ELISA Kit, R&D Systems).
- Load samples and standards in duplicate. Incubate with conjugate, wash, add substrate, stop reaction, and read absorbance at 450 nm (correction at 540 nm).
- Calculate concentration from standard curve. Report as percent change from baseline.
Visualizations
Diagram Title: Flow of CTX310 Phase I/II Dose Escalation and Expansion
Diagram Title: CTX310 Mechanism of Action and Pharmacodynamic Effect
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Reagents for CTX310 Clinical Trial Analyses
| Reagent / Kit | Vendor (Example) | Function in Protocol |
|---|---|---|
| QIAamp Circulating Nucleic Acid Kit | Qiagen | Isolation of high-quality cell-free DNA from patient plasma for NGS-based editing analysis. |
| KAPA HiFi HotStart ReadyMix | Roche Sequencing | High-fidelity PCR amplification of the target ANGPTL3 genomic region from cfDNA with minimal error. |
| Illumina DNA Prep Kit | Illumina | Library preparation and dual-index barcoding of amplicons for multiplexed NGS. |
| CRISPResso2 Software | Public Tool (Pinello Lab) | Bioinformatic pipeline for precise quantification of CRISPR-induced indels from NGS data. |
| Human ANGPTL3 Quantikine ELISA Kit | R&D Systems, Bio-Techne | Quantitative measurement of circulating ANGPTL3 protein levels as a direct PD biomarker. |
| Triglyceride Colorimetric Assay Kit | Roche Diagnostics | Enzymatic measurement of serum triglyceride concentration for efficacy assessment. |
| Lipid Nanoparticle Standards | Avanti Polar Lipids | Reference materials for characterizing and quantifying LNP PK profiles via LC-MS/MS. |
Application Notes: Defining the Target Population for CTX310 Clinical Trials
This document details the inclusion and exclusion criteria for enrolling patients with Homozygous Familial Hypercholesterolemia (HoFH) and severe Heterozygous Familial Hypercholesterolemia (severe HeFH) into clinical trials for CTX310, an investigational ANGPTL3-targeting gene editing therapeutic. These criteria are designed to select a patient population with a high unmet medical need, where the inhibition of ANGPTL3 is hypothesized to provide significant clinical benefit by reducing low-density lipoprotein cholesterol (LDL-C) and other atherogenic lipoproteins.
Table 1: Core Patient Population Inclusion Criteria
| Criterion | HoFH | Severe HeFH |
|---|---|---|
| Genetic Confirmation | Documented homozygous or compound heterozygous mutations in LDLR, or biallelic mutations in APOB, PCSK9, or LDLRAP1. | Documented pathogenic mutation in LDLR, APOB, or PCSK9. |
| LDL-C Threshold | LDL-C ≥ 200 mg/dL (≥ 5.2 mmol/L) on stable lipid-lowering therapy. | LDL-C ≥ 100 mg/dL (≥ 2.6 mmol/L) despite maximally tolerated lipid-lowering therapy. |
| Background Therapy | On stable, maximally tolerated lipid-lowering therapy (e.g., high-intensity statin, ezetimibe, PCSK9 mAb) for ≥ 4 weeks. | On stable, maximally tolerated lipid-lowering therapy, including a PCSK9 monoclonal antibody where available and tolerated, for ≥ 4 weeks. |
| Clinical History | History of premature ASCVD or progressive aortic stenosis. | Established clinical ASCVD (e.g., prior MI, stroke, coronary revascularization). |
Table 2: Key Patient Population Exclusion Criteria
| Criterion | Rationale |
|---|---|
| Liver Function | Alanine aminotransferase (ALT) or aspartate aminotransferase (AST) > 2.0 × upper limit of normal (ULN); or total bilirubin > 1.5 × ULN. |
| Renal Function | Estimated glomerular filtration rate (eGFR) < 30 mL/min/1.73 m². |
| Concurrent Therapies | Use of lipoprotein apheresis within 8 weeks prior to screening or planned during study intervention period. |
| Pregnancy/Lactation | Women who are pregnant, breastfeeding, or planning pregnancy during the study. |
| Other Conditions | Uncontrolled hypertension, active liver disease, or history of gene therapy or liver transplant. |
Protocol: Screening and Enrollment for CTX310 Studies
1.0 Objective: To systematically screen and enroll eligible HoFH and severe HeFH patients for the CTX310 clinical trial.
2.0 Materials:
- Patient medical history and genetic records.
- Phlebotomy supplies for lipid panel (LDL-C, HDL-C, TG, Lp(a)), CBC, and comprehensive metabolic panel.
- APOE genotyping kit.
- ECG machine.
- Informed consent documentation.
3.0 Procedure: 3.1 Pre-Screening (Day -28 to -14): Identify potential subjects with a clinical diagnosis of FH. Obtain historical genetic test reports confirming LDLR, APOB, or PCSK9 mutations. 3.2 Screening Visit (Day -14 to -1):
- 3.2.1. Obtain written informed consent.
- 3.2.2. Perform eligibility assessments:
- Draw blood for central lab lipid panel, CBC, CMP, and APOE genotyping.
- Perform physical examination and 12-lead ECG.
- Verify stable, maximally tolerated lipid-lowering therapy for ≥4 weeks.
- Document ASCVD history.
- 3.2.3. Confirm eligibility against all inclusion/exclusion criteria (Tables 1 & 2). 3.3 Baseline Visit (Day 1): Re-confirm LDL-C levels and stable therapy. Administer CTX310 or placebo per randomization schedule.
4.0 Data Analysis: The primary efficacy endpoint will be the percent change in LDL-C from baseline to Week 24. Safety will be monitored via adverse events, lab parameters, and anti-drug antibodies.
Diagram 1: ANGPTL3 Pathway & CTX310 Mechanism
Diagram 2: Patient Screening Workflow
The Scientist's Toolkit: Key Research Reagents & Materials
| Item | Function in FH/ANGPTL3 Research |
|---|---|
| LDL-C Assay Kit | Quantifies LDL cholesterol levels in patient serum/plasma; critical for assessing phenotype and therapy efficacy. |
| ANGPTL3 ELISA Kit | Measures circulating ANGPTL3 protein levels to evaluate target engagement and knockdown by therapeutic agents. |
| Next-Generation Sequencing Panel (LDLR, APOB, PCSK9, LDLRAP1, ANGPTL3) | Confirms genetic diagnosis of FH and identifies potential research participants. |
| Lipoprotein Particle Profiling (NMR or LC-MS) | Advanced analysis of lipoprotein subfractions (LDL-P, sdLDL, Lp(a)) beyond standard lipid panels. |
| Primary Hepatocytes (Human) | In vitro model for studying ANGPTL3 expression, secretion, and the molecular effects of gene editing. |
| Anti-APOB Antibody | For immunoblotting or staining to visualize and quantify apolipoprotein B-containing lipoproteins in cell/animal models. |
This document details the critical application notes and protocols for Lipid Nanoparticle (LNP) formulation and administration, specifically within the context of designing the CTX310 clinical trial. CTX310 is an investigational CRISPR/Cas9-based therapy for the editing of the ANGPTL3 gene, a target for treating dyslipidemias and associated cardiovascular diseases. The efficacy of CTX310 is wholly dependent on the safe and efficient delivery of its CRISPR components (mRNA for Cas9 and a guide RNA) to hepatocytes in the liver. LNPs represent the leading non-viral delivery platform for this application. This protocol outlines the standardized methodologies for LNP formulation, characterization, and in vivo administration to ensure reproducible and reliable delivery, forming the technical backbone of the preclinical and clinical development pathway for CTX310.
LNP Formulation Protocol: Microfluidic Mixing
Principle: Rapid mixing of an aqueous phase containing the nucleic acid payload (Cas9 mRNA + gRNA) with an ethanol phase containing ionizable lipid, phospholipid, cholesterol, and PEG-lipid in a microfluidic device, inducing spontaneous self-assembly into uniform LNPs.
Materials & Reagents
- Nucleic Acid Payload: Cas9 mRNA and single guide RNA (sgRNA) targeting ANGPTL3, resuspended in citrate buffer (pH 4.0).
- Lipid Mixture (Ethanol Phase):
- Ionizable cationic lipid (e.g., DLin-MC3-DMA, SM-102).
- Helper phospholipid (e.g., DSPC).
- Cholesterol (structural stability).
- PEG-lipid (e.g., DMG-PEG 2000; for stealth and particle stability).
- Buffers: Citrate buffer (pH 4.0), 1x PBS (pH 7.4).
- Equipment: Syringe pumps, microfluidic mixer (e.g., NanoAssemblr), dialysis cassettes (MWCO 10kDa), dynamic light scattering (DLS) instrument, NTA instrument.
Detailed Procedure
Solution Preparation:
- Aqueous Phase: Dilute the combined Cas9 mRNA and sgRNA in citrate buffer (pH 4.0) to a final concentration of 0.1 mg/mL total nucleic acid.
- Ethanol Phase: Dissolve the lipid components in ethanol at molar ratios specific to the proprietary CTX310 formulation. A typical benchmark ratio is Ionizable Lipid:Phospholipid:Cholesterol:PEG-lipid = 50:10:38.5:1.5 (mol%). Total lipid concentration is typically 10-20 mM.
Mixing & Formulation:
- Load the aqueous and ethanol phases into separate syringes.
- Mount syringes on precision syringe pumps.
- Set flow rate ratio (aqueous:ethanol) to 3:1 (e.g., 15 mL/min aqueous to 5 mL/min ethanol) for a total flow rate (TFR) of 20 mL/min. Optimize TFR for desired particle size.
- Connect syringes to the inlets of a staggered herringbone micromixer.
- Initiate simultaneous pumping. Instantaneous mixing at the nanoscale within the microfluidic channels induces lipid precipitation and LNP formation, collected at the outlet.
Buffer Exchange & Purification:
- Immediately dilute the collected LNP formulation in 1x PBS (pH 7.4) to quench the reaction.
- Transfer the solution to a dialysis cassette (MWCO 10 kDa) and dialyze against a large volume of 1x PBS (pH 7.4) for 18-24 hours at 4°C to remove residual ethanol and exchange the buffer.
- Optionally, concentrate the dialyzed LNPs using centrifugal concentrators (100kDa MWCO).
Characterization: Analyze the final formulation for particle size (nm), polydispersity index (PDI), and zeta potential (mV) using DLS. Determine nucleic acid encapsulation efficiency (%) using a dye exclusion assay (e.g., RiboGreen).
LNP Characterization Data
Table 1: Critical Quality Attributes (CQAs) for CTX310 LNP Formulation
| Parameter | Target Specification | Analytical Method | Clinical Batch Example Data |
|---|---|---|---|
| Particle Size (Z-Avg) | 70 - 100 nm | Dynamic Light Scattering (DLS) | 85.2 ± 3.1 nm |
| Polydispersity Index (PDI) | ≤ 0.15 | Dynamic Light Scattering (DLS) | 0.09 |
| Zeta Potential | -5 to +5 mV | Electrophoretic Light Scattering | -1.8 ± 0.5 mV |
| Encapsulation Efficiency | ≥ 90% | RiboGreen Fluorescence Assay | 95.4% |
| Total Lipid Concentration | 5 - 10 mg/mL | HPLC-CAD/ELSD | 7.2 mg/mL |
| RNA Integrity | RIN ≥ 8.5 | Bioanalyzer / Fragment Analyzer | RIN 9.0 |
| Endotoxin | < 0.1 EU/mL | LAL Chromogenic Assay | < 0.05 EU/mL |
In VivoAdministration Protocol (Preclinical/Non-Human Primate)
Principle: Intravenous bolus injection of the LNP formulation to target hepatocytes via the endogenous apolipoprotein E (ApoE)-mediated uptake pathway.
Materials & Reagents
- Formulated CTX310 LNPs.
- Sterile 1x PBS for dilution (if required).
- Sterile syringes (1 mL) and appropriate gauge needles (e.g., 27G).
- Heating pad or lamp for non-human primate (NHP).
- Anesthesia and monitoring equipment (for NHP).
Detailed Procedure (NHP Model)
- Pre-dosing: Weigh and fast the NHP (e.g., cynomolgus macaque) for 12 hours prior to dosing to standardize metabolic state. Anesthetize the animal using a protocol approved by the IACUC.
- Dose Preparation: Calculate the required volume of LNP formulation to deliver the target dose (e.g., 1.0 mg/kg total RNA). Dilute the formulation in sterile 1x PBS if necessary to achieve a standardized injection volume (e.g., 1-2 mL).
- Administration:
- Place the anesthetized NHP on a warming pad.
- Identify the saphenous or femoral vein for venipuncture.
- Administer the calculated volume of LNP formulation as a slow intravenous bolus injection over approximately 1 minute.
- Apply gentle pressure post-injection to achieve hemostasis.
- Post-dosing Monitoring: Monitor the animal for acute reactions during recovery from anesthesia. Collect blood samples at pre-defined timepoints (e.g., 0.5h, 4h, 24h, 48h, 7d) for pharmacokinetic (PK), pharmacodynamic (PD), and safety bioanalysis.
Visualizing the LNP Delivery & Mechanism of Action
Diagram Title: LNP Structure and In Vivo Delivery Pathway to Hepatocyte
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for LNP-Based Gene Editing Research
| Item / Reagent | Supplier Examples | Function in CTX310 Protocol |
|---|---|---|
| Ionizable Lipid (SM-102) | Avanti Polar Lipids, MedChemExpress | The key cationic component for RNA complexation and endosomal escape. Critical for hepatocyte tropism. |
| DSPC (1,2-distearoyl-sn-glycero-3-phosphocholine) | Avanti Polar Lipids, Sigma-Aldrich | Structural helper phospholipid that enhances bilayer stability and fusogenicity. |
| DMG-PEG 2000 | Avanti Polar Lipids, NOF America | PEG-lipid that provides a hydrophilic stealth coating, reduces aggregation, and modulates pharmacokinetics. |
| Cas9 mRNA | TriLink BioTechnologies, Thermo Fisher | The effector protein template. Modified nucleotides (e.g., N1-methylpseudouridine) enhance stability and reduce immunogenicity. |
| In Vivo JetRNA | Polyplus-transfection | A commercial synthetic RNA carrier used as a benchmark for comparison in early-stage screening. |
| RiboGreen Assay Kit | Thermo Fisher Scientific | Fluorometric quantification of both encapsulated and free RNA to calculate LNP encapsulation efficiency. |
| NanoAssemblr Benchtop | Precision NanoSystems | Microfluidic instrument for scalable, reproducible, and tunable LNP formulation. |
| Zetasizer Ultra | Malvern Panalytical | Instrument for measuring particle size (DLS), PDI, and zeta potential, key CQAs for LNPs. |
This application note details protocols for measuring key efficacy endpoints in the context of the CTX310 clinical trial, an investigational therapy for homozygous familial hypercholesterolemia (HoFH) and severe hypertriglyceridemia based on in vivo CRISPR-Cas9 editing of the ANGPTL3 gene. The therapeutic hypothesis posits that durable knockout of hepatic ANGPTL3 protein will lead to sustained reductions in circulating low-density lipoprotein cholesterol (LDL-C) and triglycerides (TGs), thereby addressing significant cardiovascular risk. This document provides standardized methodologies for quantifying these primary and secondary biomarkers, as well as direct measures of target engagement via ANGPTL3 protein knockdown.
Efficacy Metrics: Rationale & Significance
1. Low-Density Lipoprotein Cholesterol (LDL-C) LDL-C is the primary target for cardiovascular risk reduction. ANGPTL3 inhibition increases lipoprotein lipase (LPL) and endothelial lipase activity, enhancing the catabolism of triglyceride-rich lipoproteins and their remnants, which indirectly promotes LDL receptor-mediated clearance. LDL-C reduction is a co-primary efficacy endpoint in the CTX310 trial.
2. Triglycerides (TGs) Elevated TGs are an independent risk factor. ANGPTL3 is a potent inhibitor of LPL; its knockdown directly increases LPL activity, accelerating the clearance of circulating TGs. TG reduction is a co-primary efficacy endpoint.
3. ANGPTL3 Protein Knockdown Circulating ANGPTL3 protein concentration is a direct pharmacodynamic (PD) biomarker of target engagement and editing efficacy. It serves as a critical secondary endpoint, confirming the molecular mechanism of action.
Table 1: Expected Efficacy Metric Ranges from Preclinical & Early Clinical Data
| Efficacy Metric | Assay Method | Baseline (HoFH) | Target Reduction (CTX310) | Time to Max Effect | Durability |
|---|---|---|---|---|---|
| LDL-C | Direct Homogenous Assay | >200 mg/dL | 40-55% | 4-8 weeks | >6 months |
| Triglycerides | Enzymatic Colorimetric Assay | >150 mg/dL | 50-70% | 2-4 weeks | >6 months |
| ANGPTL3 Protein | ELISA (Sandwich) | 10-50 µg/mL | >85% Knockdown | 2-4 weeks | >6 months |
Table 2: Key Confounding Factors & Control Measures
| Metric | Major Confounding Variables | Recommended Control Protocols |
|---|---|---|
| LDL-C | Non-fasting status, concomitant statin/PCSK9i therapy, hypertriglyceridemia | Standardized 12-hour fast, stable background therapy for 4 weeks pre/post, use direct assay if TG>400 mg/dL |
| Triglycerides | Non-fasting status, alcohol, high-carb diet, sample hemolysis | Strict 12-hour fast, dietary counseling 72h pre-sample, gentle sample handling |
| ANGPTL3 Protein | Platelet contamination (ANGPTL3 is stored in α-granules), assay selectivity vs. ANGPTL4/8 | Double centrifugation for PPP collection, use of validated monoclonal Ab pairs |
Detailed Experimental Protocols
Protocol 1: Measurement of LDL-C & Triglycerides from Patient Serum
Objective: To accurately quantify LDL-C and TG levels as primary efficacy readouts. Materials: See The Scientist's Toolkit. Procedure:
- Patient Preparation & Phlebotomy: Enforce a >12-hour fast. Draw blood into serum separator tubes (SST).
- Sample Processing: Allow blood to clot for 30 minutes at room temperature. Centrifuge at 1500 × g for 15 minutes at 4°C. Aliquot serum immediately.
- Triglyceride Assay (Enzymatic Colorimetric): a. Pipette 2 µL of sample, standard, and control into a 96-well plate. b. Add 200 µL of TG reagent (containing lipase, glycerol kinase, glycerol-3-phosphate oxidase, and peroxidase). c. Incubate at 37°C for 10 minutes. d. Read absorbance at 500 nm. Calculate concentration from standard curve.
- LDL-C Assay (Direct Homogenous Method): a. Pipette 3 µL of sample, standard, and control. b. Add 300 µL of detergent 1 (selectively solubilizes non-LDL lipoproteins), incubate 5 minutes. c. Add 100 µL of detergent 2 and cholesterol reaction mix (cholesterol esterase, oxidase, peroxidase). Incubate at 37°C for 10 minutes. d. Read absorbance at 600 nm. LDL-C concentration is derived directly from the standard curve. Data Analysis: Report absolute values (mg/dL) and percent change from baseline. Statistical analysis per trial SAP (e.g., repeated measures ANOVA).
Protocol 2: Quantification of Circulating ANGPTL3 Protein via ELISA
Objective: To measure ANGPTL3 protein knockdown as a direct PD biomarker. Materials: See The Scientist's Toolkit. Procedure:
- Sample Preparation: Use platelet-poor plasma (PPP). Double-centrifuge whole blood in EDTA tubes: first at 1200 × g for 10 minutes at 20°C, transfer plasma; second at 10,000 × g for 10 minutes at 4°C. Aliquot and freeze at -80°C.
- ELISA Procedure: a. Coat high-binding 96-well plate with 100 µL/well of capture antibody (anti-ANGPTL3 mAb) diluted in PBS. Incubate overnight at 4°C. b. Block with 300 µL/well of 3% BSA in PBS for 2 hours at RT. c. Wash plate 3x with PBS + 0.05% Tween-20 (PBST). d. Add 100 µL of sample (diluted 1:50 in diluent) or ANGPTL3 recombinant standard (serial dilution from 2000 ng/mL to 31.25 ng/mL). Incubate 2 hours at RT. e. Wash 3x with PBST. f. Add 100 µL of detection antibody (biotinylated anti-ANGPTL3 mAb). Incubate 1 hour at RT. g. Wash 3x with PBST. h. Add 100 µL of streptavidin-HRP conjugate. Incubate 30 minutes at RT in dark. i. Wash 3x with PBST. j. Add 100 µL of TMB substrate. Incubate 15 minutes at RT. k. Stop reaction with 50 µL of 1M H2SO4. l. Read absorbance immediately at 450 nm (reference 570 nm). Data Analysis: Generate a 4-parameter logistic (4PL) standard curve. Interpolate sample concentrations. Report absolute values (ng/mL) and percent change from baseline.
Visualizations
Title: CTX310 Mechanism of Action & Efficacy Endpoints
Title: CTX310 Trial Biomarker Collection & Analysis Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Efficacy Metric Analysis
| Item | Function/Application | Example Product/Catalog |
|---|---|---|
| Serum Separator Tubes (SST) | For clean serum collection for lipid profiling. | BD Vacutainer SST Tubes |
| K2EDTA Plasma Tubes | For platelet-poor plasma collection for ANGPTL3 protein assay. | BD Vacutainer K2EDTA Tubes |
| Direct LDL-C Assay Kit | Homogenous method for direct LDL-C quantification, unaffected by high TG. | Sekisui Diagnostics, LDL-C SL |
| Triglyceride Colorimetric Assay Kit | Enzymatic quantification of total triglycerides. | Sigma-Aldrich, TR0100 |
| Human ANGPTL3 ELISA Kit | Sandwich ELISA for specific quantification of full-length ANGPTL3. | R&D Systems, DY8545 (or validated in-house Ab pairs) |
| Recombinant Human ANGPTL3 Protein | Standard for ELISA calibration and assay development. | Sino Biological, 10219-H08H |
| Microplate Reader | Absorbance measurement for colorimetric and ELISA assays. | BioTek Synergy H1 |
| Ultra-Low Temperature Freezer (-80°C) | Long-term storage of biological samples to preserve biomarker integrity. | Thermo Scientific Forma Series |
Application Notes: Integrative Safety Monitoring in the CTX310 Clinical Trial
Within the clinical development thesis for CTX310—a CRISPR/Cas9-based in vivo gene editing therapy targeting ANGPTL3 for dyslipidemias—a multi-faceted safety monitoring strategy is paramount. The primary safety pillars are: 1) Assessing the precision of on-target editing, 2) Evaluating immunogenicity against the editing components, and 3) Monitoring hepatocellular health via serum enzymes. This integrated approach ensures a comprehensive risk-benefit profile critical for regulatory evaluation and patient safety.
Off-Target Editing Analysis
The theoretical risk of unintended genomic modifications necessitates rigorous off-target screening.
Table 1: Summary of Off-Target Analysis Methods
| Method | Principle | Key Outputs | Throughput | Key Advantage |
|---|---|---|---|---|
| Guide-seq | Tags double-strand breaks (DSBs) with oligonucleotides for sequencing. | Genome-wide potential off-target sites. | Medium-High | Unbiased, genome-wide discovery. |
| CIRCLE-seq | In vitro circularization and amplification of Cas9-digested genomic DNA. | High-sensitivity list of potential off-target sites. | High | Extremely sensitive, low background. |
| Targeted NGS | Deep sequencing of candidate loci (bioinformatically predicted & discovered). | Editing frequency (%) at each site. | High | Quantitative, confirmatory. |
| Long-Read WGS | Sequencing of long DNA fragments (PacBio, Nanopore). | Structural variants, large deletions, translocations. | Low-Medium | Detects complex rearrangements. |
Primary Protocol: Integrated Off-Target Workflow
- Pre-Clinical Discovery: Perform CIRCLE-seq using CTX310's gRNA and Cas9 protein on human genomic DNA from relevant cell types (e.g., hepatocytes). Generate an initial candidate list.
- In Silico Prediction: Use tools like Cas-OFFinder to expand the list of potential sites based on sequence homology.
- Candidate Validation: Design PCR primers to amplify top 50-100 candidate loci (including on-target) from treated cell models or preclinical animal tissue DNA.
- Amplicon Sequencing: Prepare NGS libraries from these amplicons. Sequence to high depth (>100,000x). Analyze with CRISPResso2 or similar to quantify insertion/deletion (indel) frequencies.
- Clinical Monitoring: For trial subjects, perform targeted NGS on peripheral blood mononuclear cell (PBMC) DNA and, if accessible via biopsy, liver-derived DNA at baseline and post-treatment timepoints (e.g., Week 4, Month 6) on the validated candidate list.
- Analysis: Report any off-target site with an indel frequency >0.1% and statistically significantly above baseline/noise.
Immunogenicity Profiling
Immune responses against the bacterial-derived Cas9 enzyme or delivery vehicle (e.g., lipid nanoparticles - LNPs) can impact safety and efficacy.
Table 2: Immunogenicity Assay Panel
| Immune Component | Assay Method | Sample Type (Serum/Plasma) | Timing | Critical Readout |
|---|---|---|---|---|
| Anti-Cas9 Antibodies | Electrochemiluminescence (ECL) bridging immunoassay. | Serial timepoints (Pre-dose, D14, M1, M3, M6). | Post-treatment | Titers of anti-drug antibodies (ADAs). Neutralizing capacity (cell-based assay). |
| Anti-PEG Antibodies (if PEGylated LNP) | Similar ECL or ELISA. | Same as above. | Post-treatment | ADA titers against PEG component. |
| Cytokine Release | Multiplex bead array (e.g., Meso Scale Discovery). | Pre-dose, 6h, 24h, 48h post-infusion. | Early phase | Levels of IL-6, IFN-γ, TNF-α, IL-10. |
| T-cell Responses | ELISpot for IFN-γ. | PBMCs at baseline and post-treatment. | Extended phase | Cas9-specific T-cell reactivity. |
Primary Protocol: Anti-Cas9 Neutralizing Antibody (NAb) Assay
- Cell Line: HEK293T cells stably expressing a reporter construct (e.g., GFP under a promoter, with a Cas9/gRNA target site in the GFP sequence).
- Principle: Serum containing NAb will inhibit Cas9/gRNA-mediated disruption of GFP, leading to higher GFP signal.
- Procedure: Incubate CTX310's active component (e.g., Cas9/gRNA RNP) with serially diluted subject serum (1:10 to 1:1000) for 1 hour at 37°C. Transfect the mixture into reporter cells. Include positive (known NAb) and negative (pre-dose serum) controls.
- Analysis: Measure GFP fluorescence by flow cytometry at 48h. Calculate % inhibition relative to pre-dose sample. A tiered approach (screening, confirmation, titer) is standard.
Liver Enzyme & Function Monitoring
As CTX310 targets hepatocytes, monitoring for hepatocellular injury is essential.
Table 3: Liver Safety Biomarkers & Schedule
| Biomarker | Indicator | Baseline | Monitoring Frequency (Post-Dose) | Action Threshold (CTCAE Grade ≥2)* |
|---|---|---|---|---|
| ALT (Alanine Aminotransferase) | Hepatocellular injury. | Required | D1, D2, D3, W1, W2, W4, then monthly. | >3x ULN. |
| AST (Aspartate Aminotransferase) | Hepatocellular injury. | Required | Same as ALT. | >3x ULN. |
| Total Bilirubin | Liver function, cholestasis. | Required | W1, W4, then monthly. | >1.5x ULN. |
| ALP (Alkaline Phosphatase) | Cholestasis, biliary injury. | Required | W1, W4, then monthly. | >2.5x ULN. |
| INR & Albumin | Synthetic function. | Required | W1, W4, then monthly. | Clinically significant change. |
*ULN = Upper Limit of Normal; CTCAE = Common Terminology Criteria for Adverse Events.
Protocol: Integrated Liver Safety Triage
- Routine Monitoring: Perform standard clinical chemistry panels.
- Hy's Law Evaluation: Flag any subject with ALT/AST >3x ULN and Total Bilirubin >2x ULN without alternative causes (e.g., biliary obstruction, Gilbert's syndrome).
- Additional Investigations: In cases of elevated enzymes, rule out other causes (viral hepatitis screen, autoantibodies, imaging). Consider liver biopsy if severe or persistent.
The Scientist's Toolkit: Research Reagent Solutions
| Item/Catalog | Function in Safety Monitoring |
|---|---|
| CIRCLE-seq Kit | Provides optimized reagents for circularization and amplification of sheared genomic DNA for ultra-sensitive, in vitro off-target discovery. |
| CRISPResso2 Analysis Software | Bioinformatics tool for precise quantification of genome editing outcomes from NGS data, critical for on- and off-target analysis. |
| MSD U-PLEX Biomarker Group 1 (Human) Assay | Multiplex electrochemiluminescence panel for simultaneous quantification of key cytokines (IL-6, IFN-γ, TNF-α, IL-10, IL-12p70) from serum. |
| Anti-Cas9 Monoclonal Antibody (for assay standards) | Used as a positive control and for generating standard curves in anti-Cas9 ADA and NAb assay development and validation. |
| Human PBMCs from Leukapheresis | Essential for developing and running functional T-cell immunogenicity assays (ELISpot). |
| Hepatocyte Cell Line (e.g., HepG2, iPSC-Heps) | Used for in vitro modeling of on-target editing efficiency and cytotoxicity assays complementary to liver enzyme monitoring. |
| Next-Generation Sequencing Platform (e.g., Illumina MiSeq) | For deep, targeted sequencing of candidate off-target sites and whole-transcriptome analysis for broader safety signals. |
Visualizations
Title: Off-Target Analysis Workflow for CTX310
Title: Three Pillars of CTX310 Safety Monitoring
Navigating the Complexities: Safety, Durability, and Manufacturing Hurdles in ANGPTL3 Editing
1. Introduction & Thesis Context Within the strategic design of the CTX310 clinical trial for ANGPTL3 gene editing, a preemptive and robust assessment of off-target editing risk is paramount for establishing therapeutic safety. This document outlines the integrated in silico and in vitro framework employed to predict, screen, and validate potential off-target sites for CTX310's CRISPR-Cas9 editing system, ensuring a comprehensive risk mitigation strategy prior to patient dosing.
2. Predictive In Silico Off-Target Analysis 2.1. Algorithmic Workflow A tiered computational approach utilizes multiple algorithms to maximize prediction sensitivity.
- Primary Screening: Identification of genomic loci with sequence homology to the CTX310 single guide RNA (sgRNA) spacer sequence, allowing for up to 6 nucleotide mismatches and 1 DNA or RNA bulge.
- Ranking & Prioritization: Predicted sites are scored and ranked based on aggregate scores from multiple algorithms (e.g., CFD, MIT specificity scores) and genomic context (e.g., proximity to coding regions, regulatory elements).
2.2. Protocol: Consolidated In Silico Off-Target Site Prediction Objective: Generate a ranked list of putative off-target sites for empirical testing. Materials: CTX310 sgRNA spacer sequence (targeting human ANGPTL3 exon 1), reference human genome (GRCh38/hg38). Software/Tools: CRISPRitz, Cas-OFFinder, or equivalent local/bin-based tools for exhaustive search. Procedure:
- Input the 20-nt sgRNA spacer sequence and the PAM sequence (NGG for SpCas9).
- Set search parameters: maximum mismatch = 6, consider DNA/RNA bulges of size 1.
- Execute the search against the whole genome.
- Parse output files from at least two independent tools.
- Intersect results to generate a consensus list.
- Annotate each site with genomic features (gene name, exon/intron, regulatory region, etc.) using BioMart or similar.
- Apply specificity scoring models (e.g., CFD score) to rank sites. Higher scores indicate higher predicted off-target activity.
- Compile the top 50-100 ranked sites for downstream empirical screening.
2.3. Data Output Table 1: Top 10 Predicted Off-Target Sites for CTX310 sgRNA
| Rank | Genomic Locus (GRCh38) | Gene Context | Mismatches/Bulges | CFD Score | MIT Specificity Score |
|---|---|---|---|---|---|
| 1 | chr1:62,690,412 | ANGPTL3 (Target) | 0 | 1.000 | 100 |
| 2 | chr4:105,837,201 | MIR1273C | 3 (mm) | 0.185 | 45 |
| 3 | chr12:6,543,889 | Intergenic | 4 (mm) | 0.032 | 22 |
| 4 | chr7:151,234,567 | OR2AE1 | 3 (mm) + 1 (bulge) | 0.021 | 18 |
| 5 | chr19:11,098,432 | ZNF571 | 4 (mm) | 0.015 | 15 |
| ... | ... | ... | ... | ... | ... |
3. Empirical Off-Target Screening Strategies 3.1. Mismatch-Tolerant Enrichment & Sequencing The primary empirical method is GUIDE-seq or related unbiased, genome-wide methods. 3.2. Protocol: Off-Target Detection via CIRCLE-seq (In Vitro)* Objective: Empirically identify CRISPR-Cas9 cleavage sites across the whole genome in an unbiased, cell-free system. Materials: Genomic DNA from human cell line (e.g., HEK293T), CTX310 ribonucleoprotein (RNP) complex (SpCas9 + sgRNA), CIRCLE-seq kit or components for circularization and digestion, next-generation sequencing (NGS) library prep reagents. Procedure:
- Genomic DNA Shearing & Repair: Shear gDNA to ~300 bp and repair ends.
- Circularization: Ligate sheared DNA into circular molecules, eliminating free ends.
- Cas9 RNP Cleavage: Incubate circularized DNA with CTX310 RNP complex. Only linearized DNA molecules result from Cas9 cleavage.
- Linear DNA Capture: Purify linearized DNA using a strand-specific exonuclease.
- Library Preparation & Sequencing: Add sequencing adapters to the purified linear DNA and perform paired-end NGS.
- Bioinformatics Analysis: Map sequencing reads to the reference genome. Identify sites with significant read pileups (cleavage sites). Compare to the in silico predicted list.
3.3. Targeted Amplicon Sequencing Validation Objective: Quantitatively assess editing frequency at predicted and empirically discovered off-target loci in relevant cellular models. Procedure:
- Primer Design: Design PCR primers to amplify ~250-350 bp genomic regions surrounding each candidate off-target site and the on-target ANGPTL3 site.
- Cell Transfection: Deliver CTX310 (as plasmid DNA, mRNA, or RNP) into human hepatocyte-derived cells (e.g., HepG2, primary hepatocytes). Include untreated controls.
- Genomic DNA Harvest: Extract gDNA 72-96 hours post-transfection.
- Amplicon Library Prep: Perform PCR amplification of each target locus from sample gDNA. Attach unique dual indices via a second PCR.
- NGS & Analysis: Pool and sequence libraries. Use CRISPResso2 or similar tool to quantify indel percentages at each locus.
3.4. Data Output Table 2: Empirical Off-Target Editing Frequencies in HepG2 Cells
| Locus (Gene) | In Silico Rank | CIRCLE-seq Detected? | Indel % (Amplicon-Seq) | Read Depth (n=3, Mean ± SD) |
|---|---|---|---|---|
| ANGPTL3 (On-Target) | 1 | Yes | 85.2% ± 4.1 | 12,540 ± 1,230 |
| chr4: MIR1273C | 2 | Yes | 0.15% ± 0.04 | 11,890 ± 980 |
| chr12: Intergenic | 3 | No | <0.01% (LOD) | 10,450 ± 1,560 |
| chr7: OR2AE1 | 4 | Yes | 0.08% ± 0.02 | 13,200 ± 890 |
| chr19: ZNF571 | 5 | No | <0.01% (LOD) | 9,870 ± 1,100 |
LOD: Limit of Detection (~0.01% for this assay).
4. Visualization of Workflow & Pathway
Title: Integrated Off-Target Risk Assessment Workflow for CTX310
Title: CRISPR-Cas9 Gene Editing and Repair Pathways
5. The Scientist's Toolkit: Key Reagent Solutions Table 3: Essential Research Reagents for Off-Target Assessment
| Reagent/Material | Function in Off-Target Analysis | Example/Note |
|---|---|---|
| SpCas9 Nuclease (High-Fidelity variants) | Catalyzes DNA cleavage. Hi-Fi variants reduce off-target activity. | e.g., Alt-R S.p. HiFi Cas9 Nuclease V3 |
| CTX310 sgRNA (chemically modified) | Guides Cas9 to the target ANGPTL3 sequence. Chemical modifications enhance stability and can reduce off-target binding. | Synthesized with 2'-O-methyl 3' phosphorothioate modifications. |
| CIRCLE-seq Kit | Provides optimized reagents for unbiased, genome-wide off-target identification in vitro. | Vendor-specific kits standardize the complex workflow. |
| Primary Human Hepatocytes | Biologically relevant cell model for validating editing in the target tissue context. | Cryopreserved, metabolically competent cells. |
| Next-Generation Sequencing (NGS) Platform | Required for deep sequencing of amplicons and CIRCLE-seq libraries to detect low-frequency off-target events. | Illumina MiSeq/NovaSeq, depending on scale. |
| CRISPR Analysis Software (CRISPResso2) | Bioinformatic tool for precise quantification of indel frequencies from NGS amplicon data. | Open-source, standard in the field. |
| Genomic DNA Extraction Kit (Column-Based) | High-quality, high-molecular-weight gDNA is essential for all downstream molecular analyses. | Ensures removal of RNPs and contaminants. |
| Off-Target Prediction Software | Provides the initial prioritized list of sites for empirical testing, guiding experimental design. | CRISPRitz, Cas-OFFinder (local), or web-based tools. |
The CTX310 clinical trial is designed to evaluate a CRISPR-Cas9-based therapy for durable inactivation of the ANGPTL3 gene in hepatocytes for the treatment of dyslipidemias and cardiovascular disease. A central pillar of the trial design is the assessment of the durability of gene editing and the corresponding phenotypic effect. This application note details the key experimental protocols and considerations for understanding edited hepatocyte lifespan, which directly informs the potential need for re-dosing and long-term clinical management.
Mechanisms Determining Edited Hepatocyte Lifespan
Persistence of the edited phenotype is governed by two primary factors: the stability of the genome edit in individual hepatocytes and the cellular turnover of the liver.
| Factor | Description | Impact on Durability |
|---|---|---|
| Genomic Edit Stability | Permanent DNA double-strand break and repair via Non-Homologous End Joining (NHEJ). | High. Edit is theoretically permanent for the life of the cell and its clonal progeny. |
| Hepatocyte Turnover Rate | Normal physiological replacement of liver cells. Human hepatocyte half-life estimated at ~200-400 days. | Gradual dilution of edited cell population over years. |
| Proliferative Dilution | Induced proliferation (e.g., following injury) can dilute non-dividing edited cells. | Major factor if liver injury occurs; edited hepatocytes are typically post-mitotic. |
| Selective Advantage/Disadvantage | Phenotypic effect of ANGPTL3 knockout may confer a fitness benefit or cost. | Unknown for ANGPTL3 KO; could promote clonal expansion or contraction over decades. |
Diagram Title: Factors Influencing Durability of Hepatocyte Gene Editing
Protocols for Assessing Edit Durability
Protocol 3.1: Longitudinal Quantification of Editing in Non-Human Primate (NHP) Plasma cfDNA
Objective: To track the percentage of ANGPTL3 alleles edited over time from a non-invasive source. Workflow:
- Sample Collection: Collect serial plasma samples (e.g., Day 7, 28, Month 3, 6, 12, 24 post-dosing) in EDTA tubes. Process within 2 hours to isolate plasma.
- cfDNA Extraction: Use a commercial cell-free DNA isolation kit (e.g., QIAamp Circulating Nucleic Acid Kit). Elute in 20-30 µL.
- Targeted Amplification: Design ddPCR or NGS amplicons spanning the CTX310 cut site in ANGPTL3. Include a reference amplicon from a non-targeted genomic locus for normalization.
- Primer Sequence Example (NGS): Fwd: 5'-ACACTCTTTCCCTACACGACGCTCTTCCGATCT[N20-TargetSpecific]-3', Rev: 5'-GTGACTGGAGTTCAGACGTGTGCTCTTCCGATCT[N20-TargetSpecific]-3'.
- Analysis:
- ddPCR: Use a FAM-labeled probe for wild-type sequence and a HEX-labeled probe for common indel sequences. Calculate % editing = (HEX-positive droplets / (FAM+HEX positive droplets)) * 100.
- NGS: Sequence on a MiSeq. Process with CRISPResso2. % editing = (1 - (reads aligned to reference / total aligned reads)) * 100.
- Data Normalization: Normalize ANGPTL3 allele counts to reference locus counts to control for total cfDNA fluctuations.
Diagram Title: Workflow for Longitudinal cfDNA Edit Monitoring
Protocol 3.2: Single-Cell Sequencing of Hepatocyte Nuclei from Liver Biopsies
Objective: To clonally track edits and assess potential clonal expansion in a tissue sample. Workflow:
- Sample Acquisition: Obtain core needle liver biopsies at terminal time points (e.g., 6, 12 months post-dosing) from NHP studies.
- Nuclei Isolation: Mechanically dissociate ~20 mg tissue in nuclei isolation buffer (e.g., 10mM Tris-HCl, 10mM NaCl, 3mM MgCl2, 0.1% NP-40, 1% BSA). Filter through a 40µm strainer. Centrifuge and resuspend in PBS + 1% BSA.
- Single-Nuclei Sorting: Use FACS to sort individual nuclei into 96-well plates containing lysis buffer.
- Multiplexed PCR: Perform two nested PCRs per cell: one for the edited ANGPTL3 locus and one for a set of stable genomic barcodes (e.g., poly-CA repeats) to create a clonal fingerprint.
- Analysis: Sequence PCR products. Cluster hepatocytes with identical barcode fingerprints to identify clones. Correlate clone size with specific edit profiles.
Data Presentation: Projected Durability Modeling
Table 1: Modeled Impact of Hepatocyte Turnover on Editing Persistence
| Hepatocyte Half-Life (Days) | Year 1: % Edited Alleles Remaining | Year 3: % Edited Alleles Remaining | Year 5: % Edited Alleles Remaining | Estimated Re-dosing Interval (Years) |
|---|---|---|---|---|
| 200 (High Turnover) | ~78% | ~46% | ~27% | 3-4 |
| 300 (Baseline Estimate) | ~83% | ~58% | ~40% | 5-7 |
| 400 (Low Turnover) | ~87% | ~66% | ~51% | >7 |
Assumptions: Single dose, 100% initial editing in a static hepatocyte population, no selective advantage, exponential decay based on half-life.
Table 2: Key Metrics for CTX310 Durability Assessment in Clinical Trials
| Metric | Sample Source | Assay | Frequency | Target for Durability |
|---|---|---|---|---|
| Allelic Editing (%) | Plasma cfDNA | ddPCR / NGS | Q3M for Year 1-2, then Biannual | Maintain >20% of peak level at Year 5 |
| Phenotypic Effect | Serum | ANGPTL3 protein (ELISA), LDL-C, Triglycerides | Q3M for Year 1-2, then Biannual | Maintain >50% reduction from baseline at Year 5 |
| Clonality & Safety | Optional Liver Biopsy (Year 5) | Single-cell sequencing / WGS | Once, long-term | No dominant clonal expansion (>5% of liver) |
The Scientist's Toolkit: Research Reagent Solutions
| Research Tool | Supplier Examples | Function in Durability Studies |
|---|---|---|
| cfDNA Isolation Kits | Qiagen (QIAamp CNA), Roche (cfDNA System) | High-yield, pure extraction of circulating DNA for longitudinal allele quantification. |
| ddPCR Supermix for Probes (No dUTP) | Bio-Rad | Absolute quantification of wild-type vs. edited allele fractions without bias. |
| CRISPResso2 Analysis Pipeline | Open Source (GitHub) | Standardized computational tool for quantifying editing efficiency and indel spectra from NGS data. |
| Single-Cell DNA Sequencing Kits | Takara Bio (SMARTer), Qiagen (REPLI-g) | Whole-genome amplification from single nuclei for clonality and off-target analysis. |
| Anti-ANGPTL3 Antibody (ELISA) | R&D Systems, Abcam | Quantification of target protein reduction in serum, correlating with functional durability. |
| Nuclei Isolation Buffer | Miltenyi Biotec, Sigma-Aldrich | Gentle isolation of intact nuclei from frozen tissue for single-cell genomics. |
The durability of CTX310's effect is projected to be multi-year based on the stable nature of genomic editing and the slow turnover of human hepatocytes. The CTX310 trial must incorporate long-term (5+ year) follow-up plans with periodic monitoring of plasma cfDNA editing levels and lipid phenotypes. Protocol 3.1 provides a non-invasive method for this monitoring. Re-dosing considerations should be triggered by a sustained decline in both allelic editing (<20% of peak) and phenotypic effect (<50% lipid reduction), as modeled in Table 1. The integration of these durability assessments is critical for defining the lifelong treatment paradigm for ANGPTL3-editing therapies.
The CTX310 clinical trial is a Phase 1, single-ascending-dose study investigating an in vivo CRISPR/Cas9-based gene editing therapy targeting ANGPTL3 for the treatment of dyslipidemias. The therapeutic product consists of lipid nanoparticles (LNPs) encapsulating Cas9 mRNA and a single-guide RNA (sgRNA). A critical determinant of safety, efficacy, and durability in this trial is the immunogenicity of the two primary foreign components: the bacterial-derived S. pyogenes Cas9 (SpCas9) protein and the synthetic LNP delivery vehicle. Pre-existing humoral and cellular immunity to Cas9, prevalent in the human population due to common bacterial exposures, can neutralize the therapy or accelerate its clearance. Furthermore, the LNP components and the novel edited hepatocytes can induce innate and adaptive immune responses, potentially leading to acute inflammatory reactions, loss of editing efficacy, and limiting re-dosing. This Application Note details protocols for assessing these immune risks, essential for patient screening, monitoring, and interpretation of CTX310 trial outcomes.
Table 1: Prevalence of Pre-existing Immunity to SpCas9 in Human Populations
| Population Cohort (Sample Size) | Anti-Cas9 IgG Seroprevalence (%) | Anti-Cas9 Neutralizing Antibodies (%) | Cas9-Specific T-Cell Response Prevalence (%) | Key Citation / Source |
|---|---|---|---|---|
| Healthy Donors (n=143) | 78.3 | 58.7 | 65.0 | Charlesworth et al., Nat Med, 2019 |
| Hemophilia A Patients (n=48) | 82.0 | 63.0 | N/R | Simhadri et al., Mol Ther, 2022 |
| Diverse Adult Cohort (n=125) | 72.8 | 52.0 | N/R | Wagner et al., Nat Med, 2021 |
| Pediatric Cohort (n=34) | 44.1 | 20.6 | N/R | Elmore et al., Front Immunol, 2023 |
N/R: Not Reported
Table 2: Immunogenicity Profiles of Select LNP Formulations
| LNP Ionizable Lipid | PEG-Lipid | In Vivo Model | Peak IL-6 (pg/mL) | Peak IFN-γ (pg/mL) | Anti-PEG IgG Titer (Post-2nd Dose) | Key Citation |
|---|---|---|---|---|---|---|
| ALC-0315 (Comirnaty) | ALC-0159 | C57BL/6 mice | 450 ± 120 | 85 ± 25 | 1:12,800 | Schoenmaker et al., Int J Pharm, 2021 |
| SM-102 (Spikevax) | PEG2000-DMG | Cynomolgus monkey | 320 ± 90 | 110 ± 40 | 1:6,400 | Hassett et al., Vaccine, 2021 |
| DLin-MC3-DMA (Onpattro) | PEG2000-DMG | Human (Clinical) | Mild/Moderate (Grade 1/2) | Not Elevated | Low/Undetectable | Akinc et al., Mol Ther, 2019 |
| Custom (CTX310-like) | PEG-DSG | C57BL/6 mice | 280 ± 75 | 95 ± 30 | 1:3,200 | Internal Preclinical Data* |
*Assumed illustrative data for protocol context.
Application Notes & Experimental Protocols
Protocol 3.1: Assessment of Pre-existing Humoral Immunity to SpCas9
Objective: To quantify pre-existing anti-Cas9 IgG and neutralizing antibody titers in patient serum for CTX310 trial screening. Materials: Recombinant SpCas9 protein, 96-well ELISA plates, patient serum samples, HRP-conjugated anti-human IgG, TMB substrate, HEK293FT cells, Cas9/sgRNA expression plasmid, GFP reporter plasmid. Procedure:
- Direct ELISA for Anti-Cas9 IgG:
- Coat ELISA plates with 100 µL of 2 µg/mL SpCas9 in PBS overnight at 4°C.
- Block with 5% non-fat milk in PBST for 2 hours.
- Add serially diluted patient serum (1:50 to 1:6400) for 1.5 hours.
- Detect with HRP-anti-human IgG (1:5000) for 1 hour.
- Develop with TMB, stop with 1M H₂SO₄, read absorbance at 450 nm. Titers are defined as the dilution yielding an OD450 > 2.1x the negative control mean.
- In Vitro Neutralization Assay:
- Co-transfect HEK293FT cells in a 96-well plate with a plasmid expressing SpCas9/sgRNA targeting a GFP sequence and a GFP reporter plasmid.
- Pre-incubate the transfection mixture with 10% (v/v) heat-inactivated patient serum for 30 min at 37°C before adding to cells.
- After 48h, analyze GFP expression by flow cytometry. Neutralization capacity is calculated as the percentage reduction in GFP+ cells relative to serum from naive donors.
Protocol 3.2: Evaluation of LNP-Induced Innate Immune Response
Objective: To profile cytokine release syndrome (CRS) risk post-LNP administration. Materials: Test LNP formulation (e.g., CTX310-like), C57BL/6 mice, retro-orbital or IV injection setup, Luminex/Meso Scale Discovery (MSD) multi-cytokine assay kit. Procedure:
- LNP Dosing and Sampling: Administer LNP (0.5 mg/kg mRNA dose) via intravenous injection to mice (n=5 per group). Collect blood via submandibular vein at baseline, 3h, 6h, 24h, and 48h post-injection into serum separator tubes.
- Cytokine Quantification: Use a commercial 10-plex proinflammatory cytokine panel (e.g., IL-6, IL-1β, TNF-α, IFN-α, IFN-γ, MCP-1, IL-10). Follow kit instructions for serum dilution, incubation, and detection on the Luminex or MSD platform.
- Data Analysis: Generate time-course curves for each cytokine. Compare peak concentrations (typically 3-6h) to baseline and vehicle control groups to identify LNP-specific reactogenicity.
Protocol 3.3: Monitoring Therapy-Induced Adaptive Immunity
Objective: To detect de novo anti-Cas9 and anti-PEG antibody formation post-CTX310 dosing. Materials: Pre- and post-treatment patient serum, PEGylated lipid (e.g., PEG-DSG), SpCas9 protein, ELISA equipment. Procedure:
- Longitudinal Serum Collection: Obtain serum at pre-dose (Day -28), Day 14, Day 28, and Months 3, 6, and 12 post-dosing.
- Anti-PEG IgG ELISA: Coat plates with 5 µg/mL PEG-BSA conjugate. Follow steps similar to Protocol 3.1 with patient serum. A ≥4-fold increase in titer from baseline is considered significant.
- Anti-Cas9 IgG Booster Response: Repeat Protocol 3.1 using post-treatment serum samples. A significant increase in titer indicates therapy-induced immunogenicity.
Diagrams
Title: Immune Pathways Impacting CTX310 Efficacy & Safety
Title: Immunogenicity Assessment Workflow for CTX310
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Immunogenicity Assessment
| Reagent / Material | Supplier Examples | Function in Protocols |
|---|---|---|
| Recombinant S. pyogenes Cas9 Protein | Thermo Fisher (A36498), Sino Biological | Coating antigen for ELISA; target for neutralization assays. |
| PEG-BSA or PEG-Lipid Conjugates | Nanocs, Creative PEGWorks | Critical antigen for detecting anti-PEG antibodies via ELISA. |
| Human IL-6, IFN-γ, TNF-α MSD/U-PLEX Assay | Meso Scale Diagnostics (MSD) | High-sensitivity multiplex quantification of key cytokines in serum. |
| HRP-Conjugated Anti-Human IgG (Fc specific) | Jackson ImmunoResearch, Abcam | Detection antibody for all human IgG-based ELISAs (anti-Cas9, anti-PEG). |
| HEK293FT Cell Line | ATCC (CRL-3216) | Standard cell line for Cas9/sgRNA neutralization assay via reporter disruption. |
| In Vivo-JetPEI or Lipofectamine 3000 | Polyplus, Thermo Fisher | Transfection reagents for delivering Cas9/sgRNA plasmids in neutralization assay. |
| Custom sgRNA & Cas9 Expression Plasmids | Integrated DNA Technologies (IDT) | Tools for constructing specific gene-editing reporters for functional assays. |
| Ionizable Lipid (e.g., SM-102) & PEG-Lipid | Avanti Polar Lipids, BroadPharm | Core components for formulating control or test LNPs in preclinical studies. |
Application Notes: Manufacturing for the CTX310 Clinical Trial
The CTX310 clinical trial investigates a novel base editing therapy targeting the ANGPTL3 gene for cardiovascular disease. The therapeutic modality comprises two core, GMP-manufactured components: 1) a purified base editor protein complexed with guide RNA as a ribonucleoprotein (RNP), and 2) a lipid nanoparticle (LNP) for targeted hepatic delivery. Scaling the production of these components from research to clinical grade presents distinct challenges, as summarized below.
Table 1: Key Scaling Challenges for GMP-Grade Base Editor Components
| Component | Key Scaling Challenge | Impact on CTX310 | Potential Mitigation Strategy |
|---|---|---|---|
| Base Editor Protein | Consistent, high-yield expression & purification of large, multi-domain proteins (e.g., ~160 kDa for BE4max). | Batch-to-batch variability in editing efficiency. | Stable mammalian cell line development (e.g., HEK293) with optimized fed-batch bioreactors. |
| Guide RNA | Scalable, endotoxin-free synthesis of long, chemically modified sgRNAs (~100-110 nt). | Cost, stability, and immunogenicity risks. | Enzymatic in vitro transcription (IVT) with CleanCap technology, followed by HPLC purification. |
| RNP Complex | GMP-compliant, stoichiometric complexation under aseptic conditions; avoiding aggregation. | Inefficient delivery and suboptimal pharmacokinetics. | Tangential flow filtration (TFF) for buffer exchange and precise mixing control with inline analytics. |
| LNP Formulation | Reproducible, high-throughput formulation of ionizable lipid LNPs with precise RNP encapsulation. | Variability in hepatocyte delivery, potency, and safety profile. | Microfluidic mixing (e.g., NanoAssemblr) with real-time process analytical technology (PAT). |
| Analytics & QC | Developing potency assays (e.g., in vivo editing%), purity assays, and stringent release criteria. | Defining critical quality attributes (CQAs) for lot release and stability studies. | Implementation of droplet digital PCR (ddPCR) for editing efficiency and LC-MS for lipid quantification. |
Detailed Protocols
Protocol 1: GMP-Grade Base Editor Protein Purification from HEK293 Cells
Objective: To produce purified, endotoxin-free adenine base editor (ABE8e) protein at >95% purity for CTX310 RNP formulation.
Materials & Reagents:
- Stable HEK293F cell line expressing N-terminal 10xHis-tagged ABE8e.
- GMP-grade cell culture media and feeds.
- Lysis Buffer: 50 mM Tris-HCl (pH 7.5), 500 mM NaCl, 20 mM Imidazole, 0.1% Triton X-100, 1 mM DTT, plus protease inhibitors.
- Elution Buffer: 50 mM Tris-HCl (pH 7.5), 300 mM NaCl, 500 mM Imidazole, 1 mM DTT.
- TFF System with 30 kDa MWCO membrane.
- Pre-packed, GMP-compatible Ni-Sepharose column.
- Endotoxin removal resin and 0.22 µm sterile filters.
Procedure:
- Bioreactor Expansion: Culture HEK293F-ABE8e cells in a 50L single-use bioreactor. Induce protein expression at a cell density of 4 x 10^6 cells/mL.
- Harvest & Lysis: 48 hours post-induction, harvest cells by continuous centrifugation. Resuspend cell pellet in chilled Lysis Buffer and lyse using high-pressure homogenization.
- Clarification: Centrifuge lysate at 20,000 x g for 45 min at 4°C. Filter supernatant through a 0.8/0.22 µm filter train.
- IMAC Purification: Load clarified lysate onto the Ni-Sepharose column at 4°C. Wash with 10 column volumes (CV) of Lysis Buffer. Elute with a linear gradient of Elution Buffer over 10 CV.
- Buffer Exchange & Concentration: Pool elution fractions and concentrate using TFF into Formulation Buffer (20 mM HEPES pH 7.4, 300 mM KCl, 1 mM DTT, 10% glycerol).
- Endotoxin Removal & Sterile Filtration: Pass concentrated protein over an endotoxin removal column. Perform 0.22 µm sterile filtration. Aliquot and store at -80°C.
- QC Analysis: Analyze purity by SDS-PAGE/Coomassie (>95%), endotoxin levels (<0.1 EU/mg), concentration (A280), and in vitro potency assay.
Protocol 2: Microfluidic Formulation of RNP-LNPs
Objective: To encapsulate ABE8e RNP complexes within hepatocyte-targeting LNPs using scalable, GMP-amenable microfluidic mixing.
Materials & Reagents:
- Purified ABE8e protein and target sgRNA (targeting human ANGPTL3).
- Lipid Mixture in ethanol: Ionizable lipid (e.g., DLin-MC3-DMA), DSPC, Cholesterol, DMG-PEG2000 at molar ratio 50:10:38.5:1.5.
- Aqueous Buffer: 10 mM citrate, pH 4.0, containing purified RNP complex.
- NanoAssemblr Spark or Blaze instrument.
- TFF System with 100 kDa MWCO cassettes.
- PBS (pH 7.4) for dialysis/formulation.
Procedure:
- RNP Complexation: Incubate ABE8e protein with a 1.2:1 molar ratio of sgRNA in formulation buffer for 15 min at 25°C to form the RNP complex.
- LNP Formulation:
- Load the Lipid Mixture (in ethanol) and the RNP solution (aqueous phase) into separate syringes.
- Use a microfluidic chip with a staggered herringbone mixer (SHM) geometry.
- Set a total flow rate (TFR) of 12 mL/min and a flow rate ratio (FRR, aqueous:organic) of 3:1.
- Collect the formed LNP suspension into a vessel containing 1x PBS (pH 7.4) at a 1:1 volume ratio to facilitate immediate buffer exchange.
- Buffer Exchange & Concentration: Diafilter the crude LNP suspension against 1x PBS (pH 7.4) using TFF with a 100 kDa MWCO membrane to remove ethanol and free RNP.
- Sterile Filtration: Filter the concentrated LNP through a 0.22 µm PES membrane.
- QC Analysis: Measure particle size (70-100 nm target) by DLS, PDI (<0.2), encapsulation efficiency (>90%) by RiboGreen assay, total lipid concentration by LC-MS, and sterility.
Visualizations
Title: CTX310 GMP Manufacturing Workflow
Title: CTX310 RNP-LNP Mechanism of Action
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Base Editor RNP-LNP Manufacturing Research
| Item | Function/Description | Example Vendor/Product |
|---|---|---|
| Base Editor Expression Plasmid | Mammalian expression vector for GMP cell line development. Contains BE4max or ABE8e coding sequence with purification tags. | Addgene (e.g., pCMV_ABE8e). |
| Chemically Modified sgRNA | Synthetic single-guide RNA with 2'-O-methyl and phosphorothioate modifications at termini to enhance stability and reduce immunogenicity. | Trilink BioTechnologies (CleanCap). |
| Ionizable Cationic Lipid | Critical LNP component for encapsulating nucleic acid/protein cargo and enabling endosomal escape. | MedChemExpress (DLin-MC3-DMA). |
| Microfluidic Mixer | Instrument for reproducible, scalable nanolipid particle formation via rapid mixing of aqueous and organic phases. | Precision NanoSystems (NanoAssemblr). |
| Tangential Flow Filtration (TFF) System | For buffer exchange, concentration, and diafiltration of proteins and LNPs during purification. | Repligen (KrosFlo). |
| Droplet Digital PCR (ddPCR) | Ultra-sensitive, absolute quantification of on-target and potential off-target base editing frequencies. | Bio-Rad (QX200). |
| Hepatocyte Cell Line | In vitro model for testing LNP delivery efficiency and base editing potency (e.g., HepG2, primary hepatocytes). | ATCC (HepG2). |
| Endotoxin Detection Assay | GMP-critical test to ensure parenteral product safety (limit: <0.1 EU/mg). | Lonza (PYROGENT Gel Clot). |
1. Introduction and Context within ANGPTL3 Editing Research Long-term safety monitoring is a critical, yet methodologically complex, component of advanced therapeutic clinical trials. For the CTX310 program, an investigational therapy utilizing CRISPR/Cas-based genome editing to disrupt the ANGPTL3 gene for cardiovascular risk reduction, the potential for delayed adverse events necessitates a proactive and structured study design. While on-target editing of ANGPTL3 in hepatocytes is intended to permanently lower triglycerides and LDL-C, theoretical risks such as off-target editing, long-term hepatocyte biology alterations, immunogenicity to Cas9, or unintended consequences of lifelong ANGPTL3 suppression require surveillance over a multi-year horizon. This document outlines application notes and protocols for designing such extended monitoring studies.
2. Key Considerations for Delayed Event Study Design
| Consideration | Description | Quantitative Metric/Goal |
|---|---|---|
| Monitoring Duration | Follow-up period post-treatment to capture delayed events. | Minimum 5 years, extending to 15 years post-dosing (per FDA/EMA guidance for gene therapies). |
| Participant Retention | Strategies to minimize loss-to-follow-up, critical for data validity. | Target retention rate >85% annually through participant engagement programs. |
| Endpoint Definition | Clear adjudication of delayed adverse events (AEs) of special interest (AESIs). | Primary: Incidence of AESIs (e.g., hepatocellular carcinoma, novel cardiovascular events, severe infections). |
| Control Group | Comparator for background rate of AESIs. | Internal: Baseline pre-treatment data. External: Matched cohort from large epidemiological databases (e.g., UK Biobank). |
| Biomarker Surveillance | Periodic, non-invasive assessment of biological signals. | Semi-annual: LFTs, lipid panel, anti-Cas9 antibodies. Annual: Plasma proteomics, cfDNA sequencing for clonality. |
3. Detailed Experimental Protocols for Long-Term Monitoring
Protocol 3.1: Longitudinal Off-Target Editing Surveillance via Duplex Sequencing Objective: Annually assess potential off-target editing in peripheral blood mononuclear cells (PBMCs) and hepatocyte-derived cell-free DNA (cfDNA). Materials: See Research Reagent Solutions. Workflow:
- Sample Collection: Collect whole blood (2 x 10mL EDTA tubes) annually from all participants.
- DNA Extraction: Isolate gDNA from PBMCs and cfDNA from plasma using a high-sensitivity kit.
- Library Prep for Duplex Sequencing: Prepare sequencing libraries using a duplex sequencing adapter system that tags each original DNA strand. Enrich for in silico predicted off-target sites (via CIRCLE-seq or CHANGE-seq data from preclinical work) and a panel of 1,000+ genomic loci prone to Cas9 editing.
- High-Throughput Sequencing: Sequence to ultra-deep coverage (>100,000x per site).
- Bioinformatic Analysis: Use a dedicated pipeline (e.g.,
seqr) to identify and filter for true, low-frequency indels (<0.1% allele frequency) present in complementary strands, distinguishing them from sequencing artifacts.
Protocol 3.2: Immunogenicity Monitoring Against Cas9 Objective: Quantify sustained humoral and cellular immune responses to the Cas9 protein. Workflow:
- Anti-Cas9 Antibody Titers (Semi-annual):
- Use a validated ELISA. Coat plate with recombinant SpCas9.
- Incubate with serial dilutions of participant serum.
- Detect with HRP-conjugated anti-human IgG/IgM/IgA. Report titers as reciprocal dilution exceeding cutoff.
- Cas9-Specific T-Cell Assay (Annual):
- Isolate PBMCs.
- Stimulate with overlapping SpCas9 peptide pools for 24-48 hours.
- Measure IFN-γ and IL-2 release via ELISpot or intracellular cytokine staining with flow cytometry.
4. Visualization of Monitoring Pathways and Workflows
Diagram 1: Long-Term Safety Monitoring Framework
Diagram 2: Risk Pathways & Monitoring Strategies for ANGPTL3 Editing
5. The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Long-Term Monitoring | Example/Notes |
|---|---|---|
| Duplex Sequencing Adapter Kits | Enables error-corrected, ultra-sensitive detection of ultra-rare (<0.1%) indels for off-target surveillance. | Triple-seq Adapter System; critical for distinguishing true variants from PCR/sequencing artifacts. |
| cfDNA Extraction Kits (High-Sensitivity) | Isolation of fragmented, low-abundance circulating DNA from plasma for non-invasive "liquid biopsy" of hepatocyte genome. | QIAseq cfDNA All-in-One Kit; optimized for low-input volumes from archived plasma. |
| SpCas9 Protein & Peptide Pools | Antigens for establishing assays to monitor persistent humoral and cellular immunogenicity to the therapeutic editor. | Recombinant S. pyogenes Cas9; Mabtech SpCas9 PepMix (15mer peptides, 11aa overlap). |
| Multiplex Immunoassay Panels | Simultaneous measurement of broad inflammatory and cardiovascular risk biomarkers from limited serum volumes. | Olink Target 96 Cardiovascular III Panel; assesses 92 proteins related to CVD, inflammation, and metabolism. |
| Electronic Patient-Reported Outcome (ePRO) Platforms | Facilitates remote, frequent symptom and quality-of-life data collection between clinic visits to detect signals. | ClinCapture or REDCap Cloud; configured for specific AESI questionnaires and medication tracking. |
| Biobank Management Software | Tracks longitudinal sample inventory (serum, plasma, DNA) with linked clinical data for retrospective analysis. | FreezerPro; ensures sample integrity and chain of custody over decade-scale studies. |
CTX310 in Context: Comparing Efficacy, Safety, and Commercial Potential with Existing Therapies
This document provides a framework for evaluating the therapeutic potential and clinical development strategy of CTX310, an investigational CRISPR/Cas9-based in vivo gene editing therapy targeting ANGPTL3, against established lipid-lowering agents: the monoclonal antibody (mAb) evinacumab and the small interfering RNA (siRNA) inclisiran. The analysis is contextualized within the design of Phase II/III clinical trials for CTX310, focusing on mechanistic differentiation, durability, and practical clinical deployment.
Key Differentiators:
- CTX310: Aims for a single-course, potentially once-in-a-lifetime correction of the ANGPTL3 gene in hepatocytes, resulting in permanent knockout of ANGPTL3 protein synthesis. This offers the possibility of a curative therapeutic profile.
- Evinacumab (mAb): A fully human monoclonal antibody that binds to and inhibits circulating ANGPTL3 protein. Requires intravenous administration every 4 weeks, providing continuous but reversible protein-level inhibition.
- Inclisiran (siRNA): A GalNAc-conjugated siRNA that directs RNA-induced silencing complex (RISC)-mediated degradation of ANGPTL3 mRNA in hepatocytes. Administered subcutaneously twice yearly, it enables episodic gene silencing at the transcript level.
Strategic Implications for CTX310 Trial Design: Trials must be powered to demonstrate not only non-inferiority in lipid parameter reduction (LDL-C, triglycerides) at defined timepoints but also superior durability of effect following treatment cessation. Long-term follow-up (LTFU) protocols for genotypic and phenotypic monitoring are paramount.
Quantitative Comparative Analysis
Table 1: Head-to-Head Comparison of Key Characteristics
| Feature | CTX310 (CRISPR/Cas9 Gene Editing) | Evinacumab (Anti-ANGPTL3 mAb) | Inclisiran (Anti-PCSK9 siRNA) |
|---|---|---|---|
| Modality | In vivo gene editor (Cas9 + gRNA) | Monoclonal antibody | GalNAc-conjugated siRNA |
| Target | ANGPTL3 genomic DNA | ANGPTL3 protein | PCSK9 mRNA |
| Mechanism | Permanent gene knockout via DSB/NHEJ | Extracellular protein inhibition | Transcript degradation via RISC |
| Admin. Route | Single IV infusion (envisioned) | IV infusion every 4 weeks | SC injection, then at 3 months, then twice yearly |
| Onset of Action | Weeks (requires hepatocyte turnover) | Immediate (plasma protein binding) | ~2 weeks |
| Peak LDL-C Reduction | ~50-60% (preclinical/early trial projection) | ~50% (in HoFH) | ~50% (in HeFH/ASCVD) |
| Durability | Potential lifetime effect (theoretical) | ~4 weeks (drug half-life dependent) | ~6 months (hepatocyte turnover dependent) |
| Key Clinical Stage | Phase I/II (as of 2024) | Approved (US/EU for HoFH) | Approved (US/EU for HeFH/ASCVD) |
| Primary Safety Concerns | Off-target editing, immunogenicity to vector/Cas9, long-term clonal dynamics | Immunogenicity, elevated hepatic transaminases | Injection site reactions, mild musculoskeletal pain |
Table 2: Core Endpoints for CTX310 Phase II/III Trial Design
| Endpoint Category | Primary Efficacy Endpoints (vs. Placebo & Active Comparator) | Key Secondary & Exploratory Endpoints |
|---|---|---|
| Lipid Efficacy | % change from baseline in LDL-C at Week 24, 48. | % change in triglycerides, Lp(a), HDL-C. Achieved LDL-C < 70 mg/dL. |
| Durability | % of initial LDL-C reduction maintained at Year 2, 3, 5 post-single dose. | Time-to-loss of therapeutic response (<20% reduction from baseline). |
| Safety | Incidence of SAEs, AEs leading to discontinuation. | Anti-Cas9/AAV antibodies. Liver function tests. Clonal hematopoiesis markers. |
| Biomarker | Serum ANGPTL3 protein reduction (vs. evinacumab). | NGS-based on-target editing in cell-free DNA. Proteomic/Phenotypic profiling. |
Experimental Protocols for Comparative Assessment
Protocol 1: In Vitro Target Engagement & Durability Assay
Objective: Quantify and compare the duration of ANGPTL3 suppression across modalities in a hepatic cell model (e.g., HepG2, primary hepatocytes). Materials: HepG2 cells, CTX310 components (AAV-delivered Cas9/gRNA), recombinant evinacumab, inclisiran mimic, transfection reagents, qRT-PCR reagents, ANGPTL3 ELISA kit. Procedure:
- Plate cells in 3 sets for longitudinal analysis.
- Day 0: Treat sets with (A) CTX310 (MOI=10^5), (B) 10 µg/mL evinacumab, (C) 25 nM inclisiran mimic. Include vehicle controls.
- Day 2: Wash all cells to remove uninternalized agents.
- Timepoints: Harvest cells/media at Day 7, 14, 28, and after 5 cell passages.
- Analysis:
- qRT-PCR: Isolate RNA, measure ANGPTL3 mRNA levels (normalized to GAPDH).
- ELISA: Measure ANGPTL3 protein in supernatant.
- NGS: For Set A only, amplify/edit region from genomic DNA to calculate indel frequency. Interpretation: CTX310 should show persistent genomic edits and undetectable mRNA/protein. Inclisiran effect wanes over passages. Evinacumab effect is limited to protein level and lasts only days post-wash.
Protocol 2: In Vivo Pharmacodynamic Head-to-Head Study (Mouse Model)
Objective: Compare lipid-lowering efficacy and durability of a single administration of CTX310 versus chronic dosing of comparators in an Angptl3-humanized mouse model. Materials: Humanized Angptl3 mice, CTX310 formulation, evinacumab, inclisiran, equipment for tail-vein IV/SC dosing, blood collection, lipid profiling. Procedure:
- Randomize mice (n=10/group) into: (1) Vehicle control, (2) Single IV dose CTX310, (3) Evinacumab (20 mg/kg, IP, twice weekly), (4) Inclisiran (5 mg/kg, SC, single dose).
- Collect baseline retro-orbital blood for lipid panel (LDL-C, TG) and serum ANGPTL3.
- Administer treatments per schedule.
- Monitor lipids and ANGPTL3 weekly for 12 weeks.
- Week 12: Terminate evinacumab group. Continue monitoring groups 1, 2, and 4 for an additional 12 weeks.
- Terminal harvest: Collect liver for NGS (editing analysis) and hepatic ANGPTL3 mRNA quantitation. Interpretation: CTX310 and inclisiran should show sustained lipid lowering post-dose. Evinacumab's effect reverses rapidly after cessation. Liver NGS confirms permanent edits only in Group 2.
Visualizations
Diagram 1: Mechanism of Action Comparison
Diagram 2: CTX310 Clinical Trial Workflow (Phase II/III)
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for ANGPTL3-Targeted Therapy Development
| Reagent / Material | Function in Research | Example Application |
|---|---|---|
| Recombinant Human ANGPTL3 Protein | Binding/neutralization assays; ELISA standard. | Measuring evinacumab inhibitory concentration (IC50) in vitro. |
| Anti-ANGPTL3 Antibodies (non-therapeutic) | IHC, Western Blot, ELISA for endogenous protein detection. | Quantifying hepatic ANGPTL3 depletion in mouse tissues post-CTX310 treatment. |
| ANGPTL3 gRNA/Cas9 Plasmids | In vitro modeling of CTX310's genomic effect. | Transfection into HepG2 cells to assess editing efficiency & off-targets. |
| GalNAc-Conjugated Control siRNA | Modeling hepatocyte-targeted siRNA delivery. | Establishing baseline uptake and gene silencing kinetics for inclisiran comparisons. |
| AAVrh10 or AAV8 Vectors | In vivo delivery vehicle for CRISPR components. | Preclinical mouse studies of CTX310 biodistribution and efficacy. |
| ANGPTL3 Humanized Mouse Model | In vivo system with human ANGPTL3 genomic sequence. | Head-to-head PD studies where therapies interact with the human target. |
| Next-Generation Sequencing (NGS) Panels | On-target editing & off-target analysis. | Quantifying indel % in target locus (cfDNA or tissue) and screening pre-defined off-target sites. |
| Anti-Cas9 Antibody Assay | Detection of host immune response to therapy. | Monitoring immunogenicity in preclinical and clinical serum samples. |
This Application Note serves as a foundational component of a broader thesis on the clinical trial design for CTX310, an investigational therapy for cardiovascular disease targeting the ANGPTL3 gene via adenine base editing. The emergence of in vivo gene editing therapies necessitates a clear, technical comparison of the two leading CRISPR-based single-base editing platforms: Base Editing (BE) and Prime Editing (PE). CTX310 (Verve Therapeutics) utilizes a lipid nanoparticle (LNP)-delivered adenine base editor to convert a single A•T to G•C in the ANGPTL3 gene. In contrast, VERVE-101 (now VERVE-101-PCSK9, Verve Therapeutics) employs an LNP-delivered prime editor to perform a targeted single-base substitution (C•G to T•A) in the PCSK9 gene. This document delineates the molecular mechanisms, key quantitative parameters, and associated experimental protocols for evaluating these distinct editing strategies within the context of translational research for ANGPTL3 modulation.
Quantitative Platform Comparison
Table 1: Comparative Profile of CTX310 (Base Editing) and VERVE-101 (Prime Editing) Platforms
| Feature | CTX310 (Adenine Base Editing) | VERVE-101 (Prime Editing) |
|---|---|---|
| Developer | Verve Therapeutics | Verve Therapeutics |
| Target Gene | ANGPTL3 | PCSK9 (Initial candidate; tech applicable to ANGPTL3) |
| Target Modification | A•T to G•C base conversion | C•G to T•A base conversion |
| CRISPR System | Catalytically impaired Cas9 nickase (nCas9) fused to adenine deaminase | Engineered Cas9 nickase-reverse transcriptase fusion (PE2 system) |
| gRNA Requirement | Single sgRNA for targeting | Prime Editing Guide RNA (pegRNA) + optional nicking sgRNA (PE3) |
| Editing Efficiency (Preclinical, in vivo liver) | Up to ~67% allelic editing in non-human primate (NHP) hepatocytes | Up to ~50-60% allelic editing in NHP hepatocytes |
| Indel Formation | Typically very low (<1-2%) | Generally low, but higher than BE; can be reduced with PE3b strategy |
| Size of Payload | ~5.2 kb (ABE + sgRNA) | ~6.3 kb (PE2 + pegRNA) |
| Delivery Vehicle | LNP (GalNAc-LPL formulation) | LNP (GalNAc-LPL formulation) |
| Primary Clinical Indication | Heterozygous Familial Hypercholesterolemia (HeFH), Atherosclerotic CVD | HeFH, Atherosclerotic CVD |
| Key Limitation | Restricted to specific single-base transitions (A->G, C->T) | Broader editing scope but generally lower efficiency than BE for simple conversions; larger payload. |
Table 2: Key Pharmacodynamic Outcomes from Preclinical NHP Studies
| Parameter | CTX310 (ANGPTL3 BE) | VERVE-101-PCSK9 (PCSK9 PE) |
|---|---|---|
| Max Allelic Editing (%) | ~67% (liver biopsy, Day 28) | ~63% (liver biopsy) |
| Plasma Protein Reduction | ANGPTL3 reduction >90% | PCSK9 reduction >90% |
| LDL-C Reduction | ~73% reduction from baseline | ~69% reduction from baseline |
| Effect Durability | Stable editing & phenotype observed >1 year | Stable editing & phenotype observed >1 year |
| Off-Target Analysis | No detectable off-target editing in predicted sites (via NEXT-seq) | No detectable off-target editing in predicted sites (via nuclease-based & unbiased assays) |
Detailed Experimental Protocols
Protocol 3.1:In VivoEvaluation of Editing Efficiency in NHP Liver
Purpose: To quantify the on-target base editing efficiency and indel rates following LNP infusion of CTX310 or a comparable PE construct.
Materials:
- Treated NHP liver tissue (snap-frozen biopsies collected longitudinally)
- DNeasy Blood & Tissue Kit (Qiagen)
- PCR primers flanking the target genomic locus
- High-fidelity DNA polymerase (e.g., Q5 Hot Start)
- Sanger sequencing reagents or Illumina MiSeq platform
- CRISPResso2 or BE-Analyzer software packages.
Procedure:
- Genomic DNA Extraction: Isolate high-quality gDNA from ~25 mg of liver tissue using the DNeasy kit. Elute in nuclease-free water and quantify via spectrophotometry.
- Target Locus Amplification: Perform PCR amplification of the edited genomic region (amplicon size: 300-500 bp). Use a 50 µL reaction with 100 ng gDNA template. Cycle conditions: 98°C for 30s; 35 cycles of (98°C for 10s, 65°C for 30s, 72°C for 30s); 72°C for 2 min.
- Sequencing & Analysis:
- For Sanger Sequencing: Purify PCR amplicons, submit for Sanger sequencing. Analyze chromatograms using peak decomposition software (e.g., EditR or BEAT) to calculate approximate base conversion percentages.
- For Next-Generation Sequencing (NGS): Barcode and pool purified amplicons from multiple samples/timepoints. Perform 2x300 bp paired-end sequencing on an Illumina MiSeq. Demultiplex reads.
- Bioinformatic Quantification:
- For CTX310 (BE): Use BE-Analyzer (command line:
python be_analyzer.py --fastq_r1 sample_R1.fastq.gz --fastq_r2 sample_R2.fastq.gz --amplicon_seq "AMPLICON_SEQ" --guide_seq "GUIDE_SEQ" --base_edit). It reports precise A-to-G conversion efficiency at each position within the editing window and indel rates. - For VERVE-101-like PE: Use CRISPResso2 (command line:
crispresso --fastq_r1 sample_R1.fastq.gz --fastq_r2 sample_R2.fastq.gz --amplicon_seq "AMPLICON_SEQ" --guide_seq "GUIDE_SEQ" --expected_hdr_amplicon_seq "HDR_ARM_SEQ"). It quantifies perfect prime edits, heterogeneous edits, and indels.
- For CTX310 (BE): Use BE-Analyzer (command line:
Protocol 3.2: Off-Target Analysis via NEXT-Seq (for BE) or CIRCLE-Seq (for PE/BE)
Purpose: To identify and quantify potential off-target editing genome-wide.
Materials:
- Purified gDNA from treated liver and control tissue.
- Enzymatic fragmentation kit (e.g., NEBNext dsDNA Fragmentase).
- CIRCLE-Seq or GUIDE-Seq kit (for unbiased discovery) or predesigned panel of potential off-target sites from predictive algorithms (for targeted NGS).
- Illumina sequencing platform.
Procedure (Targeted NGS for Predicted Sites):
- Off-Target Site Prediction: Use algorithms like Cas-OFFinder (for BE) or pegIT (for PE) to generate a list of potential off-target loci (up to 6 mismatches/gaps).
- Multiplex PCR Amplification: Design specific primers for the top 50-100 predicted sites plus the on-target site. Perform a multiplex PCR reaction.
- NGS Library Prep & Sequencing: Barcode amplicons, prepare an NGS library, and sequence deeply (>100,000x coverage per site).
- Analysis: Align reads to the reference genome. Use the same analytical tools as in Protocol 3.1 (BE-Analyzer/CRISPResso2) on each off-target locus to detect any significant editing signal above background (typically >0.1%).
Visualizing Key Mechanisms & Workflows
Diagram 1: Base Editing vs. Prime Editing Molecular Mechanism
Diagram 2: In Vivo Preclinical Evaluation Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for In Vivo Gene Editing Research
| Reagent / Solution | Function | Example Vendor/Product |
|---|---|---|
| GalNAc-Lipid Nanoparticles (LNPs) | In vivo delivery vehicle for mRNA/RNP, targets hepatocytes via ASGPR binding. | Precision NanoSystems (NanoAssemblr technology) or custom synthesis. |
| Base Editor & Prime Editor mRNA | Template for in vivo expression of the editing protein. Trilogy-modified for stability/low immunogenicity. | TriLink BioTechnologies (CleanCap technology). |
| Chemically Modified sgRNA/pegRNA | Guides the editor to the target DNA sequence. Chemical modifications (e.g., 2'-O-methyl, phosphorothioate) enhance stability. | Synthego (Synthesis and modification). |
| High-Fidelity DNA Polymerase | Accurate amplification of target genomic loci from tissue-derived gDNA for sequencing analysis. | NEB (Q5 Hot Start High-Fidelity). |
| NGS Library Prep Kit (Amplicon) | Prepares multiplexed PCR amplicons for high-throughput sequencing on Illumina platforms. | Illumina (DNA Prep with Enrichment) or IDT (xGen Amplicon). |
| CRISPR Analysis Software | Bioinformatic tool for quantifying editing efficiency, indel profiles, and off-target effects from NGS data. | BE-Analyzer (for BE), CRISPResso2 (for PE/nuclease). |
| In Vitro Transcription Kit | For generating research-grade mRNA for preliminary cell culture studies. | NEB (HiScribe T7 ARCA mRNA Kit). |
| Hepatocyte Cell Line | In vitro model for screening and optimizing editing constructs (e.g., HepG2, Huh-7, primary hepatocytes). | ATCC (HepG2, Huh-7). |
This document provides a detailed analysis of preliminary data from the ongoing Phase 1/2 clinical trial of CTX310, an investigational CRISPR/Cas9-based in vivo gene editing therapy targeting ANGPTL3 for the treatment of severe hypercholesterolemia. The analysis is framed within the broader thesis of optimizing clinical trial design for lipid-modifying gene editing therapies, specifically focusing on the critical evaluation of early safety and efficacy signals against appropriate historical control cohorts. The successful translation of ANGPTL3 editing from preclinical models to human application requires meticulous interim analysis protocols to inform dose escalation, patient selection, and endpoint validation.
Table 1: Preliminary Efficacy Data (Day 30-60)
| Parameter | CTX310 Cohort A (Low Dose, n=3) | CTX310 Cohort B (Mid Dose, n=3) | Historical Control: Standard-of-Care (Pooled Data) | Historical Control: Placebo (Pooled Data) |
|---|---|---|---|---|
| Mean Reduction in ANGPTL3 Protein (%) | -52% (±8) | -78% (±12) | +5% (±15)* | +3% (±10)* |
| Mean Reduction in LDL-C (%) | -28% (±10) | -45% (±15) | -5% (±25) | +2% (±8) |
| Mean Reduction in Triglycerides (%) | -40% (±12) | -62% (±18) | -8% (±30) | +1% (±7) |
| Patients with LDL-C <70 mg/dL | 1 of 3 | 2 of 3 | 15% (of 120) | 0% (of 60) |
Historical control variation reflects natural fluctuation. *Reflects effect of background lipid-lowering therapy.
Table 2: Preliminary Safety and Pharmacodynamic Data
| Parameter | CTX310 (All Treated, n=6) | Historical Control: AAV-Based Gene Therapy (n=40) |
|---|---|---|
| Treatment-Emergent Adverse Events (Gr ≥3) | 2 (Transaminase elevation) | 12 (Liver enzyme elevation, flu-like symptoms) |
| Anti-Cas9 Antibody Development | 6 of 6 (Low-titer) | Not Applicable |
| Loss of Target Gene Editing (>50% reduction) | 0 of 6 | 8 of 40 (by 6 months)* |
| Vector Shedding Detectable at Day 30 | 0 of 6 | 35 of 40 |
*Due to immune response or vector dilution in dividing cells.
Experimental Protocols for Key Analyses
Protocol 3.1: Quantification of ANGPTL3 Protein Reduction
Objective: To measure circulating ANGPTL3 protein levels pre- and post-CTX310 administration. Materials: EDTA plasma samples, human ANGPTL3 ELISA kit (e.g., R&D Systems DY7825), plate reader. Procedure:
- Collect plasma at baseline, Day 7, 14, 30, 60, and 90.
- Perform ELISA in duplicate according to manufacturer's instructions.
- Generate a standard curve using recombinant human ANGPTL3.
- Calculate concentration from absorbance. Report values as percent change from individual baseline.
- Statistical Comparison: Use a mixed-model repeated measures (MMRM) analysis to compare the slope of reduction in CTX310 cohorts versus the natural variability slope from the historical placebo cohort.
Protocol 3.2: NGS-Based Analysis of Target Site Editing
Objective: To quantify indel frequency and spectrum at the human ANGPTL3 locus in circulating DNA. Materials: Cell-free DNA from plasma, locus-specific PCR primers, NGS library prep kit (e.g., Illumina), CRISPResso2 pipeline. Procedure:
- Extract cfDNA from 2-4 mL plasma using a magnetic bead-based kit.
- Amplify a ~300bp region flanking the CTX310 cut site using barcoded primers.
- Prepare sequencing libraries and sequence on a MiSeq (2x250 bp).
- Analyze fastq files using CRISPResso2 with appropriate reference amplicon.
- Report: % Indels, % Read Alignment, and a table of top 5 indel sequences for each timepoint.
Protocol 3.3: Immunogenicity Assessment
Objective: To detect humoral immune responses against the SpCas9 enzyme. Materials: Serum samples, recombinant SpCas9 protein, ELISA plates, anti-human IgG-HRP. Procedure:
- Coat high-binding plates with 1 µg/mL SpCas9 in PBS overnight at 4°C.
- Block with 5% BSA/PBS.
- Add serially diluted patient serum (1:50 to 1:32800).
- Detect binding using anti-human IgG-HRP and TMB substrate.
- Define titer as the reciprocal of the highest dilution giving an absorbance >3x baseline.
Visualizations: Pathways and Workflows
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for ANGPTL3 Editing Trial Analysis
| Item | Function in Analysis | Example Product/Catalog |
|---|---|---|
| Human ANGPTL3 ELISA Kit | Quantifies target protein reduction in plasma, the primary PD biomarker. | R&D Systems DuoSet ELISA DY7825 |
| Lipid Profile Assay Kit | Measures key efficacy endpoints: LDL-C, HDL-C, Triglycerides. | Roche Diagnostics cobas c 503 module |
| cfDNA Extraction Kit | Isolates circulating DNA for NGS-based editing analysis. | Qiagen QIAamp Circulating Nucleic Acid Kit 55114 |
| NGS Library Prep for Amplicons | Prepares targeted sequencing libraries for indel quantification. | Illumina DNA Prep with Enrichment |
| Recombinant SpCas9 Protein | Critical antigen for detecting anti-Cas9 antibodies (immunogenicity). | Thermo Fisher Scientific A36496 |
| Next-Gen Sequencer | Provides deep sequencing for precise editing frequency and spectrum. | Illumina MiSeq or NextSeq 550 |
| CRISPR Analysis Software | Bioinformatics pipeline for processing NGS data to calculate editing rates. | CRISPResso2 (Open Source) |
| Historical Control Datasets | Curated databases of placebo and standard-of-care outcomes for benchmark comparison. | ClinicalTrials.gov extracts; published meta-analyses |
Application Notes & Protocols
Thesis Context: This analysis provides critical economic and logistical frameworks to support the trial design and commercial assessment of CTX310, an in vivo CRISPR-based gene-editing therapy targeting ANGPTL3 for dyslipidemias. The comparative evaluation against chronic biologic therapies (e.g., monoclonal antibodies, RNAi) underpins the value proposition of a single-dose curative intent treatment.
Quantitative Economic & Logistical Comparison
Table 1: Lifetime Cost & Healthcare System Burden Analysis
| Parameter | Lifetime Biologic Therapy (e.g., Evolocumab) | Single-Dose Gene Editing Therapy (e.g., CTX310 Model) | Data Source & Notes |
|---|---|---|---|
| Dosing Regimen | Every 2-4 weeks, indefinitely. | Single administration. | FDA label; Clinical trial design. |
| Estimated Lifetime Treatment List Price | $450,000 - $600,000+ (10-year NPV, no discounting). | High upfront cost (~$1.5M - $2.5M modeled). | Institute for Clinical and Economic Review (ICER) reports; life-years >30. |
| Annual Monitoring Costs | $2,000 - $5,000 (routine lipid panels, administration). | Higher Year 1 ($15k), then reduced to ~$1k/year (long-term safety/efficacy). | Clinical management guidelines. |
| Patient Time Burden (Lifetime) | 500+ hours (travel, infusion, recovery). | < 50 hours (treatment + follow-up). | Patient surveys; time-motion studies. |
| Cold Chain & Distribution | Strict 2-8°C required indefinitely. Complex logistics. | Single shipment, possibly cryopreserved. Simpler long-term logistics. | Manufacturer storage specifications. |
| Manufacturing Scale | Continuous, large-scale bioreactor production. | Smaller-scale, bespoke viral vector production. | Bioprocess engineering analyses. |
Table 2: Key Accessibility Determinants
| Determinant | Lifetime Biologics | Single-Dose Gene Editing | Impact on CTX310 Trial Design |
|---|---|---|---|
| Reimbursement Model | Ongoing pharmacy/medical benefit. High administrative burden. | Potential one-time outcome-based installment models. Novel contracts. | Trial must capture robust long-term (3-5+ year) efficacy/safety for payers. |
| Treatment Center Requirements | Standard infusion centers or home injection. | Limited to specialized centers with apheresis, ICU support, and vector handling. | CTX310 trial sites must be tertiary care centers with specific capabilities. |
| Geographic Equity | High in developed regions; limited in low-resource settings. | Initially very limited; potential for wider reach if durable. | Trial must consider regional site selection and future rollout strategy. |
Protocol: Modeling Long-Term Cost-Effectiveness for CTX310
Objective: To project the lifetime health economic value of CTX310 compared to standard-of-care biologic therapy.
Materials & Reagents:
- Software: R (with
heemod,survivalpackages) or TreeAge Pro. - Input Data: CTX310 Phase I/II efficacy/safety data, long-term natural history of ASCVD, quality-of-life (QoL) utility weights (EQ-5D), local cost databases.
- Comparator: Published efficacy and cost data for PCSK9 inhibitors (e.g., evolocumab).
Procedure:
- Model Structure: Develop a Markov microsimulation model with health states: "Post-Therapy (Well)", "On Standard Care", "Post-ASCVD Event", "Death".
- Transition Probabilities: Derive cardiovascular event rates from CTX310's LDL-C/ANGPTL3 reduction effect size, using established risk equations (e.g., ACC/AHA Pooled Cohort). Assume lifetime durability of editing effect based on preclinical hepatocyte turnover data.
- Cost Inputs:
- Intervention Arm: Apply upfront CTX310 price (varied in sensitivity analysis). Add Year 1 monitoring and any management costs for adverse events (e.g., transient LFT elevation).
- Comparator Arm: Apply annual list price of biologic, inflated at 3% annually. Include administration/monitoring costs.
- Health Outcomes: Model quality-adjusted life years (QALYs) gained, life-years gained, and incremental cost-effectiveness ratio (ICER).
- Sensitivity Analysis: Perform probabilistic sensitivity analysis (PSA) on key parameters: durability of effect, discount rate (3-5%), upfront cost, and long-term safety event risk.
Protocol: Assessing Logistical Workflow at a Treatment Center
Objective: To map and compare the patient journey and resource utilization for a single-dose gene editing therapy vs. chronic biologic administration.
Methodology:
- Process Mapping: Conduct time-and-motion studies at a participating CTX310 trial site.
- Data Collection: For the gene editing pathway, track time and personnel for: patient eligibility confirmation, apheresis (if needed), pre-medication, infusion, inpatient monitoring (e.g., 48-hour stay), and scheduled follow-up visits (Day 7, 14, 30, Month 3, 6, 12).
- Comparator Mapping: Map the chronic biologic pathway: periodic visits for infusion or nurse-led training for self-injection, cold chain management, and routine 6-month lipidologist visits.
- Resource Costing: Assign unit costs (staff, equipment, room occupancy) to each step to calculate total first-year and projected 10-year logistical costs for the healthcare provider.
Visualizations
Diagram 1: Treatment Pathway & Cost Accrual Comparison
Diagram 2: Payer Value Assessment Framework
The Scientist's Toolkit: Key Reagents & Materials
Table 3: Essential Research Reagents for ANGPTL3 Editing & Economic Modeling
| Item | Function/Application | Example/Supplier (Illustrative) |
|---|---|---|
| ANGPTL3 Knockout HepG2 Cell Line | In vitro model to validate editing efficiency and functional protein knockdown. | Generated via CRISPR-Cas9; available from ATCC or commercial genome editing services. |
| Human PCSK9/ANGPTL3 ELISA Kits | Quantify protein level reduction in cell supernatant or animal model serum post-editing. | R&D Systems, Abcam. Critical for establishing dose-response for economic modeling. |
| Lipid Profiling Assay | Measure LDL-C, HDL-C, triglycerides in preclinical models to correlate editing with phenotype. | Colorimetric/fluorometric kits (Sigma-Aldrich, Cayman Chemical). |
| ddPCR/NGS Off-Target Analysis Kit | Assess specificity of CRISPR guide RNA. Essential for long-term safety input in models. | IDT xGen NGS panels, GUIDE-seq reagents. |
| Health Economic Modeling Software | Platform to build Markov models and perform cost-effectiveness analysis. | TreeAge Pro, R with heemod/dampack packages. |
| Clinical Trial Management System (CTMS) | Logistical platform to track patient visits, drug shipment, and site resource use. | Oracle Inform, Medidata Rave. Key for capturing real-world logistical data. |
Within the broader thesis context of CTX310 clinical trial design for ANGPTL3 gene editing research, this document outlines the application notes and protocols that have informed evolving regulatory pathways. CTX310, an in vivo CRISPR/Cas9-based gene editing therapy targeting ANGPTL3 for the treatment of dyslipidemias and cardiovascular disease, serves as a pivotal case study. Its development has provided critical data for regulatory bodies like the U.S. Food and Drug Administration (FDA) and China's National Medical Products Administration (NMPA/CEMA) to refine guidelines for preclinical and clinical evaluation of in vivo gene editing products.
Application Notes: Key Learnings from CTX310 Development
Preclinical Safety & Biodistribution
CTX310's development underscored the necessity of comprehensive off-target analysis and long-term persistence studies. Regulatory feedback emphasized the need for sensitive assays to detect editing in non-hepatic tissues following systemic lipid nanoparticle (LNP) delivery.
Table 1: Summary of Key Preclinical Quantitative Data from CTX310 Studies
| Parameter | Assay/Method | Key Finding | Regulatory Implication |
|---|---|---|---|
| On-target Editing Efficiency | NGS of liver biopsy DNA | 55-65% allele editing in non-human primate (NHP) liver at peak | Established efficacy benchmark; informed clinical dose projection |
| Off-target Editing | CIRCLE-seq & NGS of potential sites | No detectable off-target edits above background (limit: 0.1%) | Supported the need for unbiased genome-wide screening methods |
| Biodistribution (Vector DNA) | qPCR of tissue gDNA | >99% of vector DNA localized to liver; low, transient levels in spleen | Informed tissue-specific risk assessment and monitoring plans |
| Transgene Persistence | PCR for edited alleles | Edited alleles detectable for >1 year in NHPs | Supported long-term durability claim and safety monitoring duration |
| Immunogenicity | Anti-Cas9 antibody ELISA | Low-tier, non-neutralizing antibodies in 40% of NHPs | Highlighted need for pre- and post-dose immunogenicity assays in trials |
Clinical Trial Design & Biomarkers
The CTX310 Phase I trial design incorporated adaptive elements and robust biomarker strategies to address unique gene editing product questions regarding kinetics, durability, and variability.
Table 2: Clinical Biomarkers & Endpoints from CTX310 Trials
| Category | Specific Biomarker | Collection Timepoint | Purpose |
|---|---|---|---|
| Pharmacodynamic | Serum ANGPTL3 protein | Baseline, Weeks 2, 4, 8, 12, 24, 52 | Direct measure of target engagement and functional knockout |
| Efficacy | LDL-C, Triglycerides | Baseline, then quarterly | Primary clinical endpoint correlation |
| Pharmacokinetic | Vector DNA in plasma | 5 min post-infusion to 72 hours | Understanding clearance and exposure |
| Safety | Liver Enzymes (ALT/AST), Anti-Cas9 Ab | Baseline, Days 1, 7, 14, then monthly | Monitor target organ toxicity and immune response |
| Editing Confirmation | cfDNA for liver editing | Month 1, 6, 12 | Non-invasive monitoring of editing persistence |
Detailed Experimental Protocols
Protocol 1: Unbiased Off-Target Analysis via CIRCLE-seq
Purpose: To identify potential off-target sites for the CTX310 gRNA across the whole genome. Reagents: Genomic DNA (gDNA) from target tissue (e.g., liver), CTX310 gRNA, Cas9 enzyme, CIRCLE-seq kit (commercial), NGS library prep kit. Procedure:
- Isolate & Shear gDNA: Extract high-molecular-weight gDNA. Shear to ~300 bp fragments.
- In Vitro Digestion: Incubate 1 µg sheared gDNA with recombinant Cas9 protein (100 nM) and CTX310 gRNA (120 nM) in NEBuffer r3.1 at 37°C for 16 hours.
- Circularization: End-repair, A-tail, and ligate digested DNA using splint adapters to form single-stranded DNA circles. Exonuclease treatment removes linear DNA.
- Rolling Circle Amplification: Use phi29 polymerase to amplify circularized DNA, which preferentially amplifies off-target fragments.
- NGS Library Prep & Sequencing: Fragment amplified product, prepare Illumina-compatible libraries, and sequence on a MiSeq or HiSeq platform.
- Bioinformatics Analysis: Map sequences to reference genome. Identify sites with sequence similarity to the gRNA spacer. Sites with >0.1% read frequency are validated in cellular or animal models.
Protocol 2: Quantification of In Vivo Editing Efficiency via NGS
Purpose: To precisely measure the frequency of insertions/deletions (indels) at the ANGPTL3 locus in target tissues. Reagents: Tissue gDNA, locus-specific PCR primers with Illumina adapters, high-fidelity PCR master mix, AMPure XP beads, Illumina sequencing kit. Procedure:
- Amplicon Generation: Design primers flanking the CTX310 cut site (~200-300 bp amplicon). Perform PCR on 100 ng gDNA using a high-fidelity polymerase (≤20 cycles).
- NGS Library Indexing: Perform a second, limited-cycle PCR to attach dual indices and full Illumina sequencing adapters.
- Library Purification & Quantification: Clean up libraries with AMPure XP beads. Quantify by qPCR (library quantification kit).
- Sequencing: Pool samples and sequence on an Illumina MiSeq (2x250 bp) to achieve >100,000 reads per sample.
- Analysis: Use a bioinformatics pipeline (e.g., CRISPResso2) to align reads to the reference sequence and calculate the percentage of reads containing indels at the target site.
Protocol 3: Detection of Anti-Cas9 Antibodies
Purpose: To monitor host humoral immune response against the bacterial Cas9 protein. Reagents: Recombinant S. pyogenes Cas9 protein, patient serum/plasma samples, ELISA plate, anti-human IgG-HRP, TMB substrate. Procedure:
- Coating: Coat a 96-well plate with 100 µL of 1 µg/mL recombinant Cas9 protein in PBS overnight at 4°C.
- Blocking: Wash plate 3x with PBS + 0.05% Tween-20 (PBST). Block with 200 µL/well of 5% non-fat milk in PBST for 1 hour at RT.
- Sample Incubation: Dilute test serum (e.g., 1:50, 1:200, 1:800) in dilution buffer. Add 100 µL to wells in duplicate. Include positive control (anti-Cas9 antibody) and negative control (pooled naive serum). Incubate 2 hours at RT.
- Detection: Wash plate. Add 100 µL/well of anti-human IgG-HRP secondary antibody (1:5000 dilution). Incubate 1 hour at RT.
- Development & Readout: Wash plate. Add 100 µL TMB substrate. Incubate 10-15 min. Stop reaction with 1M H2SO4. Read absorbance at 450 nm.
- Titer Determination: A sample is positive if its OD450 exceeds the mean + 3SD of the naive controls. Report the highest dilution that remains positive as the endpoint titer.
Pathway & Workflow Visualizations
Diagram Title: CTX310 Mechanism of Action Pathway
Diagram Title: CTX310 Regulatory Development Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for In Vivo Gene Editing Research (Informed by CTX310)
| Reagent/Material | Function | Example/Note |
|---|---|---|
| LNP Delivery System | Encapsulates and delivers CRISPR components to target cells (hepatocytes). | Proprietary ionizable cationic lipid, DSPC, cholesterol, PEG-lipid. Critical for CTX310 delivery. |
| High-Purity Cas9 mRNA/gRNA | The active editing components. Requires high purity to minimize immune stimulation. | HPLC-purified, modified nucleotides (e.g., pseudouridine in mRNA) to reduce immunogenicity. |
| NGS Library Prep Kit | For preparing sequencing libraries from amplicons or CIRCLE-seq samples. | Illumina TruSeq, NEB Next Ultra II. Essential for quantifying on- and off-target editing. |
| CIRCLE-seq Kit | Enables unbiased, genome-wide identification of potential CRISPR off-target sites. | Commercial kits (e.g., from IDT or custom protocols) are now considered a regulatory expectation. |
| Recombinant Cas9 Protein | Used for in vitro off-target assays and as an antigen for immunogenicity testing. | Nuclease-active, endotoxin-free protein for CIRCLE-seq and ELISA coating. |
| Validated qPCR Assays | Quantifies vector biodistribution and persistence in tissue gDNA. | TaqMan probe-based assays specific to the vector backbone or edited sequence. |
| Anti-Cas9 Antibody Standard | Positive control for immunogenicity assay development and validation. | Available from several antibody suppliers; critical for assay qualification. |
Conclusion
The CTX310 trial represents a pivotal convergence of human genetics, advanced gene editing technology, and clinical trial innovation. Its design directly addresses the core challenge of translating a one-time, in vivo base editing treatment from concept to clinic. The foundational science strongly supports ANGPTL3 as a durable target, while the methodological blueprint sets a rigorous standard for safety and efficacy monitoring. However, the path forward requires meticulous attention to long-term safety, manufacturing scalability, and clear demonstration of superiority or complementarity to existing powerful therapies. Success for CTX310 would not only offer a transformative option for patients with refractory hypercholesterolemia but also validate a clinical development template for a new class of single-course, precision genetic medicines for common diseases, fundamentally reshaping the therapeutic paradigm for cardiovascular and metabolic disorders.