Decoding the Immune Response to Cas9: A Comprehensive Guide to Epitope Mapping and Immunodominant HLA Epitopes for Safer Gene Therapies
This article provides a comprehensive resource for researchers and drug developers on the critical intersection of Cas9 immunogenicity, epitope mapping, and HLA-restricted immune responses.
Decoding the Immune Response to Cas9: A Comprehensive Guide to Epitope Mapping and Immunodominant HLA Epitopes for Safer Gene Therapies
Abstract
This article provides a comprehensive resource for researchers and drug developers on the critical intersection of Cas9 immunogenicity, epitope mapping, and HLA-restricted immune responses. We explore the foundational biology of Cas9 as a foreign antigen, detailing established and emerging methodologies for identifying its B-cell and T-cell epitopes. The guide systematically addresses common challenges in epitope prediction and experimental validation, offering optimization strategies for deimmunizing Cas9 variants. Finally, we present a comparative analysis of validation techniques and discuss how this knowledge directly informs the design of safer, next-generation CRISPR-Cas9 therapeutics with reduced immunogenic risk, crucial for successful clinical translation.
Understanding Cas9 Immunogenicity: The Why and How of Immune Recognition in Gene Editing
This whitepaper analyzes the immunogenicity of the CRISPR-associated protein 9 (Cas9), primarily derived from Streptococcus pyogenes (SpCas9), within the framework of ongoing research on Cas9 epitope mapping and the identification of immunodominant Human Leukocyte Antigen (HLA) epitopes. The bacterial origin of Cas9 renders it a foreign antigen in humans, potentially triggering pre-existing and adaptive immune responses that pose significant challenges for in vivo therapeutic applications, such as gene therapy and gene editing.
Immunogenicity arises from two principal sources: pre-existing humoral immunity (neutralizing antibodies) and cell-mediated immunity (Cas9-specific CD4+ and CD8+ T-cells). These are directed against bacterial Cas9 epitopes due to widespread human exposure to common bacterial species like S. pyogenes.
Table 1: Prevalence of Pre-existing Immunity to SpCas9 in Human Populations
| Study Cohort (Reference) | % Seropositive (IgG) | % T-cell Response Positive | Key Epitopes Identified | HLA Association |
|---|---|---|---|---|
| Healthy Adults (Charlesworth et al., 2019) | 58% | 78% | Multiple CD4+ & CD8+ | HLA-DR, HLA-A*02:01 |
| Pediatric Cohort (Simhadri et al., 2018) | ~40% | 65% | Not Fully Mapped | Various |
| In Silico Analysis (Wagner et al., 2019) | N/A | N/A | Predicted Dominant (e.g., SpCas910-22) | HLA-DRB104:01, HLA-A02:01 |
Detailed Experimental Protocols for Epitope Mapping
Protocol:In VitroT-cell Activation Assay for Cas9 Epitope Discovery
Objective: To identify immunodominant CD4+ and CD8+ T-cell epitopes within the SpCas9 protein. Materials:
- Peripheral Blood Mononuclear Cells (PBMCs) from healthy human donors.
- Overlapping peptide library (15-mer peptides, 11-aa overlap) spanning the full SpCas9 sequence.
- Positive controls: Anti-CD3 antibody, PHA.
- Negative control: DMSO (peptide solvent).
- Detection reagents: ELISpot kits for IFN-γ, IL-2, or flow cytometry antibodies for activation markers (CD69, CD137) and intracellular cytokines. Procedure:
- Isolate and cryopreserve PBMCs from donor blood using Ficoll density gradient centrifugation.
- Thaw PBMCs and rest overnight in complete RPMI-1640 medium.
- Plate PBMCs (2-5 x 10⁵ cells/well) in 96-well plates pre-coated with anti-IFN-γ antibody (for ELISpot).
- Stimulate cells with individual Cas9 peptides (1-10 µg/mL), positive, and negative controls.
- Incubate for 24-48 hours at 37°C, 5% CO₂.
- Develop ELISpot plates per manufacturer's protocol to visualize cytokine-secreting cells. For flow cytometry, add brefeldin A/GolgiStop after 2 hours, incubate 4-6 more hours, then stain for surface and intracellular markers.
- Analyze. A response is considered positive if peptide-stimulated wells have at least 2-fold more spot-forming units (SFUs) or cytokine+ T-cells than the mean of negative control wells and exceed a predefined threshold (e.g., 50 SFUs/10⁶ cells).
Protocol: HLA Epitope Prediction and Binding Affinity Assays
Objective: To computationally predict and biochemically validate HLA class I and II epitopes within Cas9. Materials:
- Recombinant HLA proteins (specific alleles like HLA-A*02:01).
- Radio- or fluorescence-labeled indicator peptides.
- Cas9-derived candidate peptides (9-10mers for Class I, 15-20mers for Class II).
- Detergent-solubilized cell extracts expressing single HLA alleles. Procedure (Competitive Binding Assay):
- Prediction: Use netMHCpan or IEDB analysis resource to predict high-affinity Cas9 peptides for common HLA alleles.
- Incubation: Incubate purified HLA molecules with a fixed concentration of labeled indicator peptide and varying concentrations of unlabeled Cas9 test peptide.
- Separation: Separate HLA-peptide complexes from free peptide using gel filtration, affinity capture, or a scintillation proximity assay.
- Quantification: Measure the amount of bound labeled peptide. The concentration of Cas9 peptide required to displace 50% of the labeled peptide is calculated as the IC₅₀.
- Validation: Peptides with IC₅₀ < 500 nM are considered high-affinity binders and prioritized for cellular validation (Protocol 3.1).
Visualization of Immune Recognition Pathways
Title: Cellular Immune Response to Cas9 Antigen
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Cas9 Immunogenicity Research
| Item | Function/Application |
|---|---|
| SpCas9 Overlapping Peptide Library | A set of synthetic peptides spanning the entire protein sequence for comprehensive in vitro T-cell epitope screening. |
| PBMCs from Characterized Donors | Primary human immune cells with known HLA haplotypes, essential for ex vivo immune response assays. |
| Recombinant HLA Allele-Specific Tetramers | Fluorescent MHC-peptide complexes used to identify and isolate Cas9-specific T-cells via flow cytometry. |
| Anti-Human IFN-γ ELISpot Kit | Standardized assay to quantify Cas9-reactive T-cells based on cytokine secretion at the single-cell level. |
| NetMHCpan Prediction Server | Computational tool for in silico prediction of Cas9-derived peptide binding affinity to specific HLA alleles. |
| Cas9-Specific IgG ELISA Kit | Pre-coated plate assay to detect and quantify pre-existing anti-Cas9 antibodies in human serum/plasma. |
| HLA-typed Humanized Mouse Models | In vivo models (e.g., NSG-A2) expressing human HLA molecules to study Cas9-specific immune responses. |
The clinical application of genome editing tools, particularly CRISPR-Cas9 systems, represents a paradigm shift in therapeutic development. However, their bacterial origin renders them foreign to the human immune system. This technical guide explores the critical imperative of preexisting immunity against therapeutic vectors and proteins, focusing on its profound impact on efficacy and safety. This discussion is framed within a broader research thesis centered on Cas9 epitope mapping and the characterization of immunodominant HLA epitopes. The ubiquitous exposure to Streptococcus pyogenes (the source of SpCas9) and related bacteria in the human population has led to widespread preexisting humoral and cellular immunity, which can neutralize therapeutic vectors, reduce engraftment, and trigger adverse inflammatory responses. Understanding and circumventing this immunity is a non-negotiable prerequisite for successful clinical translation.
Quantifying Preexisting Immunity: Prevalence Data
Recent seroprevalence and cellular immunity studies provide a quantitative basis for the clinical concern. The data below summarizes key findings from recent investigations.
Table 1: Prevalence of Preexisting Immunity to Common CRISPR-Cas Orthologs in Human Populations
| Cas9 Ortholog | Source Bacterium | Seroprevalence (IgG) | T-Cell Reactivity Prevalence | Primary Study & Year |
|---|---|---|---|---|
| SpCas9 | Streptococcus pyogenes | 58-78% | 67-85% (CD8+) | Wagner et al., Nature Med, 2021 |
| SaCas9 | Staphylococcus aureus | ~24% | ~46% (CD4+) | Charlesworth et al., Nature Med, 2019 |
| AsCas12a | Acidaminococcus sp. | ~21% | ~34% (CD8+) | Ferdosi et al., Nat Comm, 2023 |
| LbCas12a | Lachnospiraceae bacterium | <10% | ~15% (estimated) | Simhadri et al., Mol Ther, 2022 |
Table 2: Clinical Consequences of Preexisting Immunity in Early-Phase Trials
| Therapeutic Platform | Immune Target | Observed Impact | Outcome Metric Change |
|---|---|---|---|
| AAV-based Gene Therapy | AAV Capsid | Reduced transgene expression | >90% reduction in high-titer subjects |
| Ex Vivo CRISPR-Edited Cell Therapy | SpCas9 Protein | Enhanced clearance of edited cells | 2-3 fold faster clearance vs. naive |
| In Vivo mRNA-LNP CRISPR | Cas9/SgRNA RNP | Elevated inflammatory cytokines (IL-6, TNF-α) | Grade 2-3 adverse events correlated with high titer |
Core Experimental Protocols for Epitope Mapping and Immunogenicity Assessment
Protocol: HLA-Peptidomics for Cas9 Immunodominant Epitope Discovery
Objective: To directly isolate and sequence Cas9-derived peptides presented by HLA class I and II molecules on antigen-presenting cells.
- Cell Culture & Antigen Presentation: Immortalized human B-cells (e.g., C1R cells) stably expressing a single HLA allotype are transfected with a SpCas9 expression vector or pulsed with recombinant SpCas9 protein.
- HLA Complex Immunoprecipitation: After 24-48h, cells are lysed in mild detergent. HLA-I (using antibody W6/32) and HLA-II complexes are separately immunoprecipitated from the lysate.
- Peptide Elution & Cleanup: Bound peptides are eluted using 10% acetic acid at 72°C, separated from the HLA heavy chain by ultrafiltration (10-kDa cutoff).
- LC-MS/MS Analysis: Peptides are analyzed by nano-flow liquid chromatography coupled to tandem mass spectrometry. Data-dependent acquisition is used.
- Bioinformatics: MS/MS spectra are searched against the human proteome plus the SpCas9 sequence using software (e.g., MaxQuant). Peptides uniquely mapping to Cas9 are identified, and binding affinity to the expressing HLA is validated in silico (NetMHCpan, NetMHCIIpan).
Protocol: T-Cell Activation Assay for Validating Epitope Immunodominance
Objective: To functionally validate the capacity of predicted/mapped epitopes to activate CD4+ or CD8+ T-cells from seropositive donors.
- Donor PBMC Isolation: Peripheral blood mononuclear cells (PBMCs) are isolated from healthy donors with confirmed anti-Cas9 antibodies via density gradient centrifugation.
- Epitope Pooling: Predicted immunodominant 15-mer (CD4+) or 9-10-mer (CD8+) peptides are synthesized. Peptides are pooled into matrices for deconvolution.
- In Vitro Stimulation: PBMCs are cultured with peptide pools (1 µg/mL per peptide) in the presence of co-stimulatory antibodies (anti-CD28/CD49d) and IL-2 for 10-14 days.
- ELISpot/Intracellular Cytokine Staining (ICS):
- IFN-γ ELISpot: Restimulated cells are added to IFN-γ-coated plates with individual peptides. Spot-forming units (SFUs) are counted to identify reactive peptides.
- ICS: Cells are restimulated with peptides in the presence of brefeldin A, stained for surface CD4/CD8 and intracellular IFN-γ/TNF-α, and analyzed by flow cytometry. The frequency of cytokine-positive T-cells quantifies reactivity.
Visualizing Key Concepts and Workflows
Diagram 1: Impact of Preexisting Immunity on CRISPR Therapy
Diagram 2: Workflow for Mapping Immunodominant Cas9 Epitopes
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Preexisting Immunity & Epitope Research
| Reagent / Material | Supplier Examples | Function in Research |
|---|---|---|
| Recombinant Cas9 Proteins (Sp, Sa, As) | Sino Biological, Origene, ABclonal | Used as antigens in ELISA, Luminex, and T-cell stimulation assays to measure humoral and cellular immunity. |
| HLA Typed PBMCs & Sera | PrecisionMed, AllCells, BioIVT | Provide diverse, characterized human immune cells and antibody sources from healthy and diseased donors for ex vivo immunogenicity studies. |
| PepMix Peptide Pools (Cas9) | JPT Peptide Technologies | Overlapping 15-mer peptide libraries spanning the entire Cas9 protein for comprehensive T-cell epitope screening via ELISpot/ICS. |
| MHC Tetramers/Pentamers (Custom) | ProImmune, MBL International | Fluorescently labeled multimers loaded with specific Cas9 epitopes to directly identify and isolate antigen-specific T-cells by flow cytometry. |
| Anti-Human IFN-γ ELISpot Kit | Mabtech, Cellular Technology Ltd. | Gold-standard functional assay to quantify the frequency of Cas9-reactive T-cells from PBMC samples upon peptide stimulation. |
| HLA Class I/II Immunoprecipitation Kits | BioLegend, Thermo Fisher | Enable the isolation of peptide-HLA complexes from cell lysates for subsequent mass spectrometric analysis (HLA-peptidomics). |
| Pseudotyped Lentivirus (Cas9/SaCas9) | VectorBuilder, GeneCopoeia | Used in in vitro neutralization assays to model how patient sera can inhibit the transduction efficiency of Cas9-delivery vectors. |
Within the framework of Cas9 epitope mapping and immunodominant HLA epitope research, a precise understanding of B-cell and T-cell epitope dichotomy is fundamental. This review delineates the structural and functional distinctions between these epitope classes, emphasizing the indispensable role of HLA presentation in shaping adaptive immune responses, a critical consideration for therapeutic protein and vaccine development.
Defining B-cell vs. T-cell Epitopes
B-cell (Linear & Conformational) Epitopes
B-cell epitopes are specific regions of an antigen recognized by the B-cell receptor (BCR) or a secreted antibody. They are categorized as:
- Linear/Sequential: Comprise continuous amino acid sequences.
- Conformational/Discontinuous: Formed by spatially adjacent residues from different segments of the folded polypeptide chain.
T-cell Epitopes (Linear Only)
T-cell epitopes are short, linear peptide fragments derived from proteolytic processing of the antigen. They are not recognized in their native form but are presented by Major Histocompatibility Complex (MHC/HLA) molecules on the surface of antigen-presenting cells (APCs) for recognition by the T-cell receptor (TCR).
Table 1: Core Distinctions Between B-cell and T-cell Epitopes
| Feature | B-cell Epitope | T-cell Epitope |
|---|---|---|
| Recognizing Receptor | B-cell Receptor (BCR) / Antibody | T-cell Receptor (TCR) |
| Antigen Form Recognized | Native, folded 3D structure | Processed linear peptide |
| Requires HLA Presentation | No | Yes, mandatory |
| Typical Size | 5-25 amino acids (conformational can be larger) | 8-15 amino acids (HLA class I), 13-25 (HLA class II) |
| Nature | Linear or Conformational | Exclusively Linear |
| Key Function | Direct neutralization, Opsonization | Cell-mediated killing (CD8+), Helper functions (CD4+) |
The Central Role of HLA Presentation in T-cell Epitope Selection
HLA Class I and II Pathways
T-cell epitope presentation follows two primary pathways, dictating the immune effector response.
HLA Polymorphism and Epitope Immunodominance
HLA genes are highly polymorphic, leading to individual-specific "peptide-binding motifs." An epitope presented by a common HLA allele may become immunodominant—eliciting a strong, focused T-cell response. In Cas9 research, identifying immunodominant epitopes presented across diverse HLA alleles is crucial to predict and mitigate unwanted immune responses.
Table 2: Quantitative Analysis of HLA Restriction and Epitope Prediction (Recent Data)
| Parameter | HLA Class I | HLA Class II | Notes / Source |
|---|---|---|---|
| Typical Binding Core Length | 9-mer (8-12) | 15-mer (13-25) | Anchored by 2-3 key residues |
| Number of Canonical HLA Alleles | ~20,000 (IPD-IMGT/HLA DB v3.54) | ~8,000 (IPD-IMGT/HLA DB v3.54) | Reflects extreme polymorphism |
| Epitope Prediction Algorithm AUC* | 0.85 - 0.95 (NetMHCpan-4.1) | 0.75 - 0.85 (NetMHCIIpan-4.0) | *Area Under Curve for performance |
| Reported Immunodominant Cas9 Epitopes | 3-5 per isoform (e.g., SpCas9) | 2-4 per isoform (e.g., SpCas9) | Identified via in vitro assays |
Experimental Protocols for Epitope Mapping in Cas9 Research
Protocol: In Silico Prediction of T-cell Epitopes
Objective: To computationally predict potential immunogenic T-cell epitopes within the Cas9 protein sequence.
- Sequence Retrieval: Obtain the full amino acid sequence of the Cas9 variant (e.g., UniProt ID Q99ZW2 for S. pyogenes Cas9).
- Allele Selection: Choose a panel of HLA alleles representative of the target population (e.g., HLA-A02:01, DRB103:01).
- Prediction Execution: Run the sequence through prediction servers:
- For HLA-I: Use NetMHCpan-4.1 or IEDB MHC-I binding predictor. Submit sequence, select allele list, and use default parameters (predicted IC50 < 50 nM considered strong binder).
- For HLA-II: Use NetMHCIIpan-4.0 or IEDB MHC-II binding predictor. Specify appropriate chain (DR, DP, DQ).
- Data Aggregation: Compile lists of strong- and weak-binding peptides. Rank based on predicted binding affinity and proteasomal cleavage score (for HLA-I).
Protocol: In Vitro Validation Using ELISpot
Objective: To experimentally validate CD4+ or CD8+ T-cell responses to predicted Cas9 epitopes.
- Peptide Synthesis: Synthesize predicted 15-mer (HLA-II) or 9-mer (HLA-I) peptides, often as overlapping peptide libraries.
- PBMC Isolation: Isolate Peripheral Blood Mononuclear Cells (PBMCs) from healthy or pre-exposed donors.
- Cell Stimulation: Plate PBMCs in an IFN-γ pre-coated ELISpot plate. Add individual peptides or pools. Include positive (PHA) and negative (media) controls.
- Incubation & Development: Incubate for 24-48 hours. Follow manufacturer's protocol to add detection antibodies, streptavidin-enzyme conjugate, and substrate to visualize spots.
- Analysis: Enumerate spots (each representing an epitope-specific, cytokine-secreting T-cell) using an automated ELISpot reader. Responses are typically considered positive if spot count exceeds mean + 2-3 SD of negative control and a threshold (e.g., >50 SFC/10^6 PBMCs).
Protocol: HLA Peptide Affinity Assay
Objective: To biochemically measure the binding affinity of a candidate peptide for a specific HLA molecule.
- Purified HLA Incubation: Incubate purified, recombinant HLA monomer with a fluorescently labeled reference peptide and a titrated concentration of the unlabeled candidate Cas9 peptide in a buffer containing a protease inhibitor cocktail.
- Competition: Allow competition for the HLA binding groove to reach equilibrium (typically 24-48 hours at room temperature).
- Separation & Detection: Separate bound from unbound peptide using size-exclusion chromatography or a capture assay. Measure the fluorescence of the HLA-bound fraction.
- IC50 Calculation: Plot the decreasing signal of bound reference peptide against the log concentration of the candidate peptide. Calculate the IC50 (concentration inhibiting 50% of reference peptide binding). An IC50 < 500 nM is generally considered a high-affinity binder.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Epitope Mapping Studies
| Item | Function in Research | Example/Catalog Consideration |
|---|---|---|
| Overlapping Peptide Libraries | Covers entire protein sequence for unbiased epitope mapping; used in ELISpot/T-cell stimulation. | JPT PepMix, 15-mers with 10-aa overlap. |
| Recombinant HLA Monomers | Essential for direct, quantitative peptide-binding affinity assays. | Immune Monitoring HLA Class I & II Tetramers. |
| IFN-γ ELISpot Kits | Gold-standard for quantifying epitope-specific T-cell frequency and functionality. | Mabtech Human IFN-γ ELISpot PLUS kit. |
| Antigen-Presenting Cells | Engineered cell lines (e.g., T2 for HLA-I, CHO for HLA-II) to study presentation. | T2 (HLA-A2:01), Raji (HLA-DR). |
| Tetramer Reagents | Fluorescently labeled peptide-HLA complexes for direct staining and sorting of epitope-specific T-cells. | NIH Tetramer Core Facility reagents. |
| Proteasome Inhibition Cocktail | Used in cellular assays to differentiate HLA-I pathway processing. | MG-132, Epoxomicin. |
| HLA-Typed PBMCs | Critical for assessing epitope presentation across diverse genetic backgrounds. | Commercial vendors (e.g., Stemcell) or IRB-approved donor collections. |
The precise discrimination between B-cell and T-cell epitopes, governed by the rules of HLA presentation, forms the mechanistic bedrock for de-risking biologics like Cas9 nucleases. Integrating in silico prediction with robust in vitro validation protocols allows researchers to map immunodominant regions systematically. This approach is paramount for engineering evolved proteins with reduced immunogenic risk, ultimately enabling safer therapeutic applications.
Abstract Immunodominance defines the hierarchical preference of T-cell responses for a limited subset of potential epitopes derived from complex antigens, a phenomenon strictly governed by HLA presentation and T-cell receptor (TCR) repertoire. Within the thesis framework of Cas9 epitope mapping and its implications for therapeutic gene editing, understanding immunodominance is critical. This guide details the molecular determinants, experimental methods for deconvoluting epitope hierarchies, and their direct relevance to predicting and mitigating anti-Cas9 immune responses in clinical applications.
1. Core Determinants of Epitope Hierarchy Immunodominance is not a random occurrence but the product of a multi-step filtration process. The key determinants are:
- Antigen Processing & HLA Binding Affinity: The primary filter. Epitopes must be generated by the proteasome, transported via TAP, and bind with high affinity to the HLA molecule. Affinity is typically measured as IC50 (nM).
- TCR Repertoire: The precursor frequency of naïve T-cells bearing TCRs capable of recognizing the pHLA complex dictates response magnitude.
- Epitope Abundance & Stability: The density and longevity of pHLA complexes on the APC surface correlate with immunogenicity.
- Immuno-informatic Predictions: Computational tools predict binding affinity, but in vitro and ex vivo validation is mandatory.
Table 1: Quantitative Metrics for Dominant vs. Subdominant Epitopes
| Parameter | Immunodominant Epitope | Subdominant/Cryptic Epitope |
|---|---|---|
| Predicted HLA Binding Affinity (IC50) | <50 nM | >500 nM |
| Measured HLA Binding Stability (t½) | > 6 hours | < 2 hours |
| Precursor Frequency (Tetramer+ CD8+ T-cells) | ~10⁻⁵ – 10⁻⁶ | <10⁻⁷ |
| Ex Vivo ELISpot Response (SFU/10⁶ PBMCs) | >500 | <100 |
| Role in Anti-Cas9 Immune Response | Primary driver, clinically relevant | Minor or undetectable role |
2. Experimental Protocols for Epitope Hierarchy Mapping These protocols are central to the thesis work on mapping Cas9-specific T-cell epitopes.
2.1. In Silico Prediction & HLA Binding Assay
- Objective: Identify candidate epitopes from SpCas9 protein sequence.
- Protocol:
- Use NetMHCpan (v4.1) and IEDB consensus tools to predict 9-10mer peptides binding to prevalent HLA alleles (e.g., A*02:01, B*07:02).
- Synthesize top-scoring peptides (typically IC50 < 500 nM).
- Perform a competitive HLA-binding assay: Incubate purified HLA molecules with a labeled indicator peptide and serial dilutions of the unlabeled candidate peptide.
- Measure displacement; calculate IC50. Peptides with IC50 < 50 nM are high-affinity binders.
2.2. Epitope-Specific T-cell Detection (Tetramer Staining)
- Objective: Quantify precursor frequency of epitope-reactive T-cells.
- Protocol:
- Generate PE- or APC-conjugated HLA tetramers loaded with the candidate peptide.
- Isolate PBMCs from donor blood.
- Stain PBMCs with tetramer, anti-CD3, anti-CD8, and viability dye for 30 min at 4°C.
- Analyze by flow cytometry. A positive population is defined as CD3+CD8+tetramer+ and distinct from negative controls.
2.3. Functional Validation (IFN-γ ELISpot)
- Objective: Confirm immunogenicity and rank epitope potency.
- Protocol:
- Coat ELISpot plates with anti-IFN-γ capture antibody overnight.
- Seed PBMCs or isolated CD8+ T-cells (2-5 x 10⁵ per well) with peptide-pulsed autologous APCs (or directly with peptide).
- Incubate for 24-48 hours at 37°C.
- Develop with biotinylated detection antibody, streptavidin-ALP, and BCIP/NBT substrate.
- Count spot-forming units (SFU) using an automated reader. Dominant epitopes elicit strong responses without prior in vitro expansion.
3. Visualization of Determinants and Workflow
Determinants of Immunodominant T-cell Epitopes
Experimental Workflow for Epitope Mapping
4. The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Immunodominance Research
| Reagent / Material | Function & Application |
|---|---|
| Recombinant HLA Class I Monomers | Core component for constructing tetramers and performing in vitro binding assays. |
| Peptide Libraries (SpCas9-derived) | Defined pools of predicted epitopes for high-throughput screening of T-cell responses. |
| Fluorochrome-Conjugated Streptavidin (PE, APC) | Used with biotinylated HLA monomers to generate multivalent tetramers for flow cytometry. |
| Anti-IFN-γ ELISpot Kit (Mabtech) | Gold-standard for quantifying epitope-specific T-cell responses via cytokine secretion. |
| T-cell Activation/Maturation Markers (CD137, CD69) | Antibodies to detect early and late activation of T-cells upon epitope encounter. |
| HLA-typed PBMCs from Healthy Donors | Critical ex vivo substrate for validating epitope immunogenicity and establishing hierarchy. |
| NetMHCpan & IEDB Analysis Resource | Computational platforms for initial epitope prediction and narrowing candidate lists. |
5. Relevance to Cas9 Epitope Mapping For therapeutic Cas9 delivery, pre-existing or induced immunodominance towards certain epitopes presents a major hurdle. A dominant, conserved epitope presented by a common HLA allele could lead to immune clearance of edited cells or adverse events. Systematic mapping of the immunodominant hierarchy across common HLA alleles enables:
- Risk Stratification: Identifying patients with high-response HLA alleles.
- Protein Engineering: Deimmunizing Cas9 by mutating anchor residues in dominant epitopes without affecting function.
- Therapeutic Monitoring: Using tetramers for dominant epitopes to track immune responses in clinical trials.
Conclusion Defining immunodominance is a quantitative, multi-parametric exercise essential for predicting adaptive immune responses. Within Cas9 research, applying these principles allows for the proactive management of immunogenicity, guiding the engineering of safer gene-editing therapeutics and informing patient-specific treatment strategies.
The clinical application of CRISPR-Cas9 gene editing is contingent on overcoming immune recognition. Cas9 proteins, commonly derived from Streptococcus pyogenes (SpCas9), contain peptide sequences that can be presented by Human Leukocyte Antigen (HLA) molecules, potentially triggering cytotoxic T-cell responses. High-prevalence HLA alleles, such as those from the HLA-DR locus (Class II) and HLA-A*02:01 (Class I), are of paramount importance in Cas9 epitope mapping research. Their global frequency dictates the proportion of the population at risk for immune-mediated clearance of Cas9-expressing cells or adverse events. This whitepaper synthesizes current data on the global distribution of key HLA alleles and details the experimental protocols for mapping immunodominant Cas9 epitopes restricted by these alleles, a critical step in developing safer, immunologically stealth gene therapies.
Global Population Coverage of Key HLA Alleles
The following tables summarize the allele frequency data for high-prevalence HLA Class I and Class II alleles across major geographic populations, based on recent analyses from the Allele Frequency Net Database and population genomics studies.
Table 1: Global Frequency of Selected Prevalent HLA Class I Alleles
| HLA Allele | Global Avg. Frequency (%) | European Frequency (%) | East Asian Frequency (%) | African Frequency (%) | Admixed American Frequency (%) | South Asian Frequency (%) |
|---|---|---|---|---|---|---|
| A*02:01 | 15.2 | 24.8 | 13.5 | 7.1 | 16.3 | 11.9 |
| A*01:01 | 7.8 | 14.2 | 2.1 | 3.5 | 6.0 | 6.5 |
| A*03:01 | 9.1 | 13.5 | 3.8 | 8.9 | 7.4 | 10.2 |
| B*07:02 | 7.5 | 10.9 | 3.5 | 5.8 | 8.1 | 8.0 |
| C*07:01 | 14.3 | 15.8 | 11.2 | 18.5 | 16.9 | 13.1 |
Table 2: Global Frequency of Selected Prevalent HLA Class II Haplotypes/Alleles
| HLA Allele/Haplotype | Global Avg. Frequency (%) | European Frequency (%) | East Asian Frequency (%) | African Frequency (%) | Admixed American Frequency (%) | South Asian Frequency (%) |
|---|---|---|---|---|---|---|
| DRB107:01-DQA102:01-DQB1*02:02 | 8.5 | 12.1 | 1.5 | 10.3 | 14.2 | 4.8 |
| DRB115:01-DQA101:02-DQB1*06:02 | 6.2 | 11.3 | 7.8 | 2.1 | 5.5 | 4.0 |
| DRB103:01-DQA105:01-DQB1*02:01 | 5.9 | 9.8 | 0.5 | 6.5 | 4.1 | 3.2 |
| DRB104:01-DQA103:01-DQB1*03:02 | 3.7 | 6.5 | 4.2 | 0.8 | 4.8 | 2.1 |
| DRB1*09:01 | 3.5 | 1.0 | 15.2 | 0.2 | 2.1 | 2.5 |
Table 3: Cumulative Population Coverage for Common HLA Allele Combinations
| Allele Set | Phenotype Coverage - Global (%) | Phenotype Coverage - Europe (%) | Phenotype Coverage - East Asia (%) | Phenotype Coverage - Africa (%) |
|---|---|---|---|---|
| HLA-A02:01, A01:01, A*03:01 | ~52% | ~75% | ~32% | ~45% |
| HLA-DRB107:01, DRB115:01, DRB1*03:01 | ~45% | ~65% | ~20% | ~40% |
| Combined Set (All 6 above) | ~85% | ~95% | ~65% | ~80% |
Experimental Protocols for Cas9 Epitope Mapping
In SilicoPrediction of HLA-Restricted Cas9 Epitopes
Protocol:
- Sequence Retrieval: Obtain the full amino acid sequence of the Cas9 protein of interest (e.g., SpCas9, UniProt ID Q99ZW2).
- Peptide Fragmentation: Perform an in silico digest into overlapping peptides (typically 9-mers for Class I, 15-mers for Class II).
- HLA Binding Prediction: Utilize neural network-based algorithms.
- For Class I (e.g., HLA-A02:01): Use NetMHCpan-4.1 or MHCflurry 2.0. Input the 9-mer peptides and the specific HLA allele. A predicted rank percentile threshold of <0.5% is commonly used for strong binders.
- For Class II (e.g., HLA-DRB107:01): Use NetMHCIIpan-4.0. Input 15-mer peptides. A predicted rank threshold of <2% is often used for strong binders.
- Immunogenicity Prediction: Filter predicted binders through tools like PRIME 2.0 or DeepImmuno to rank their potential to elicit T-cell responses.
- Output: Generate a prioritized list of candidate immunodominant epitopes for experimental validation.
Ex VivoT-Cell Activation Assay (ELISpot)
Protocol:
- Peripheral Blood Mononuclear Cell (PBMC) Isolation: Collect whole blood from HLA-typed donors (e.g., HLA-A*02:01+). Isolate PBMCs via density gradient centrifugation (Ficoll-Paque).
- Peptide Stimulation: Resuspend candidate peptides (predicted epitopes) in DMSO and dilute in serum-free media. Plate PBMCs (2-5 x 10^5 cells/well) in an IFN-γ pre-coated ELISpot plate. Add peptides (final conc. 1-10 µg/mL). Include positive controls (PHA or CEF peptide pool) and negative controls (DMSO vehicle).
- Incubation: Incubate plates for 24-48 hours at 37°C, 5% CO₂.
- Detection: Follow manufacturer's protocol (e.g., Mabtech human IFN-γ ELISpot kit). Typically involves washing, addition of biotinylated detection antibody, followed by streptavidin-ALP and BCIP/NBT substrate.
- Analysis: Enumerate spots using an automated ELISpot reader. A response is considered positive if the mean spot-forming units (SFU) in the test well exceeds the mean of negative controls by a predefined threshold (e.g., 2-fold) and is >50 SFU/10^6 PBMCs.
Confirmatory HLA Restriction Analysis (MHC Deblocking)
Protocol:
- Setup: Using PBMCs from a responsive donor, set up duplicate ELISpot or intracellular cytokine staining (ICS) reactions with the immunodominant peptide.
- Antibody Blocking: Add HLA-specific blocking monoclonal antibodies to the test wells.
- For Class I restriction: Add anti-HLA-ABC (W6/32) or allele-specific antibody (e.g., anti-A2, BB7.2).
- For Class II restriction: Add anti-HLA-DR (L243), anti-HLA-DQ (SPVL3), or anti-HLA-DP (B7/21).
- Control: Include isotype-matched control antibodies.
- Assay Completion: Complete the ELISpot/ICS protocol as described above.
- Interpretation: A significant reduction (>50%) in cytokine response (SFU or % cytokine+ cells) in the presence of the specific HLA-blocking antibody, but not the isotype control, confirms the HLA restriction element for the epitope.
Cas9 Epitope Mapping and HLA Restriction Workflow
HLA Class II Mediated Immune Recognition of Cas9
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Reagents for Cas9 HLA Epitope Mapping Research
| Reagent / Material | Function & Application | Example Product / Note |
|---|---|---|
| Recombinant Cas9 Protein | Source antigen for direct in vitro antigen presentation assays or for immunizing HLA-transgenic mice. | Commercial SpCas9 (nuclease active or inactive). Ensure high purity (>95%) and low endotoxin. |
| Synthetic Peptide Libraries | Overlapping peptides spanning the Cas9 sequence for screening predicted epitopes in T-cell assays. | Custom 15-mer peptides overlapping by 10-11 aa (for Class II); 9-mer libraries (for Class I). Purity >70%. |
| HLA-Typed PBMCs | Primary human cells from donors with specific, high-prevalence HLA alleles for ex vivo immunogenicity testing. | Obtain from commercial biorepositories (e.g., STEMCELL Tech) or clinical collaborators with IRB approval. |
| IFN-γ ELISpot Kit | Gold-standard for detecting antigen-specific T-cell responses via cytokine secretion at the single-cell level. | Mabtech Human IFN-γ ELISpotPRO kit; includes pre-coated plates, antibodies, and reagents. |
| HLA-Blocking Antibodies | Monoclonal antibodies used to confirm HLA restriction of identified epitopes (MHC deblocking assay). | Anti-HLA-ABC (W6/32 clone), Anti-HLA-DR (L243 clone), and allele-specific antibodies (e.g., BB7.2 anti-A2). |
| HLA Tetramers/Pentamers | Fluorescently labeled MHC-peptide complexes for direct staining and isolation of epitope-specific T-cells. | Custom ProImmune MHC Pentamers or Tetramers for validated Cas9 epitopes. Critical for monitoring immune responses. |
| HLA-Transgenic Mice | In vivo models expressing human HLA alleles (e.g., HLA-A2.1/DR1) for immunogenicity and safety studies. | Available from Jackson Laboratory (e.g., B6.Cg-H2-Ab1 |
| Immunoinformatics Software | Platforms for in silico prediction of HLA binding, antigen processing, and T-cell epitopes. | NetMHCpan-4.1, NetMHCIIpan-4.0, IEDB Analysis Resource, PRIME 2.0. |
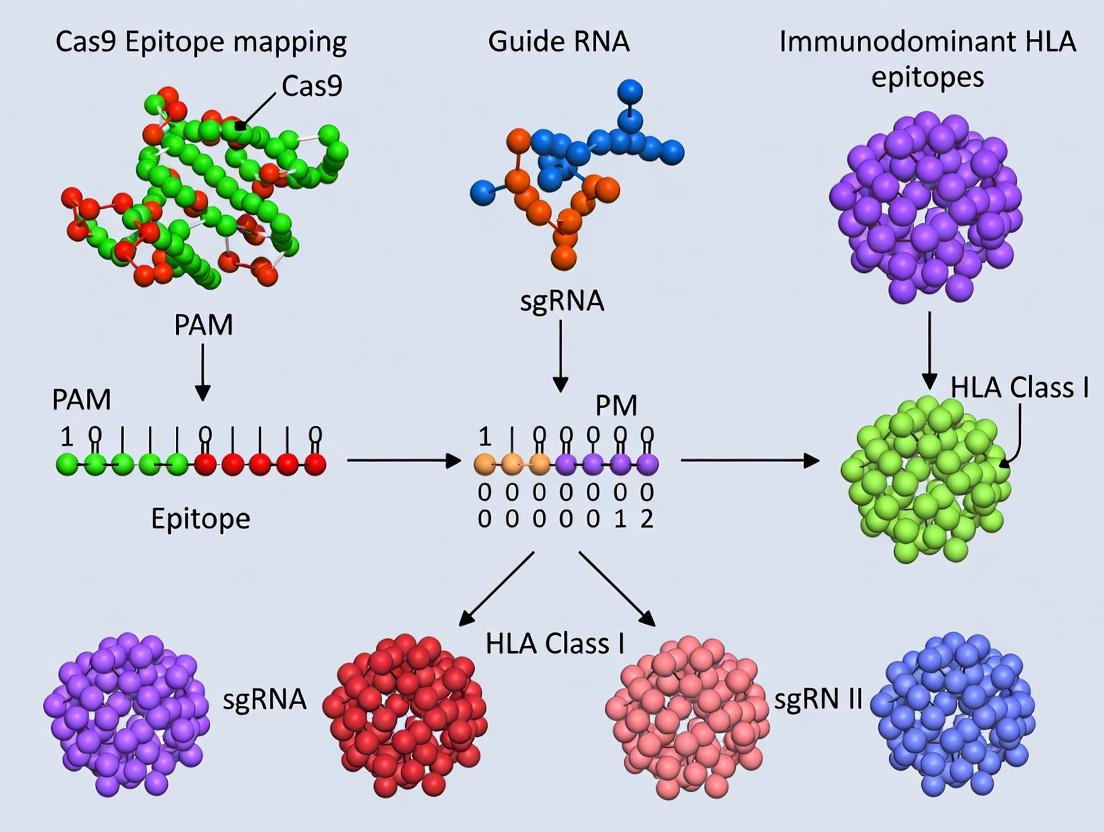
Mapping the Immune Landscape: Proven Techniques for Cas9 Epitope Discovery and Analysis
This guide is framed within a broader research thesis focused on identifying non-immunogenic regions for in vivo Cas9 protein delivery. The central challenge is that Cas9, derived from Streptococcus pyogenes, contains immunodominant epitopes that can trigger HLA class II-restricted CD4+ T cell responses, leading to antibody formation and clearance of engineered cells. In silico prediction of HLA class II epitopes is therefore a critical first step for de novo immunogenicity screening and the rational design of engineered, "stealth" Cas9 variants with reduced immunogenic potential.
2.1 The IEDB Analysis Resource Consensus Approach The Immune Epitope Database (IEDB) provides a suite of tools, with its consensus method for MHC-II being a standard. It aggregates predictions from multiple constituent methods (e.g., NetMHCIIpan, NN-align, SMM-align, Combinatorial Library) to produce a single percentile rank. A lower rank indicates higher predicted binding affinity.
Table 1: Key Characteristics of Featured Prediction Tools
| Tool Name | Core Algorithm | Primary Output | Key Strength | Consideration for Cas9 Mapping |
|---|---|---|---|---|
| IEDB Consensus | Ensemble of algorithms | Percentile Rank | Robust, community standard | Good for initial broad screening of peptides. |
| NetMHCIIpan 4.3 | Artificial Neural Networks | %Rank, nM Affinity | Pan-specific for HLA-DR/DQ/DP | High accuracy for diverse alleles; essential for diverse donor cohorts. |
| Deep Learning Models (e.g., DeepLigand, MHCnuggets) | Convolutional/Recurrent Neural Networks | Binding Score | Captures complex sequence patterns | Emerging tool; may identify non-canonical binding cores. |
2.2 NetMHCIIpan: A Pan-Specific Methodology NetMHCIIpan is the leading pan-specific predictor, capable of predicting for any HLA-DR, DQ, and DP allele using artificial neural networks trained on eluted ligand and binding affinity data. Version 4.3+ offers improved accuracy for rare alleles, critical for population-wide immunogenicity risk assessment.
2.3 Emerging Deep Learning Models Deep learning architectures (CNNs, RNNs, Transformers) process amino acid sequences as multidimensional feature maps, learning hierarchical patterns indicative of binding without relying on pre-defined peptide cores. They are trained on expansive MS-eluted ligand datasets.
Detailed Experimental Protocol forIn SilicoCas9 Epitope Mapping
This protocol outlines a comprehensive prediction workflow suitable for a thesis project on Cas9 immunogenicity.
Step 1: Protein Sequence and Allele Selection.
- Input: Obtain the full-length amino acid sequence of S. pyogenes Cas9 (UniProt ID: Q99ZW2).
- Allele Set: Select HLA-II alleles representative of the target population. A minimal set covering common DRB1 allotypes is: DRB1*01:01, *03:01, *04:01, *07:01, *08:01, *11:01, *13:01, *15:01. Include DQ and DP alleles for comprehensive analysis.
Step 2: In Silico Peptide Generation.
- Method: Perform an in silico digest using a sliding window. For MHC-II, a 15-mer window is standard, as it accommodates the core 9-mer binding register flanked by extensions.
- Software/Code: Use
IEDB's Peptide Fragmenteror a simple Python script (e.g.,[peptide for i in range(len(sequence)-14): peptide = sequence[i:i+15]]).
Step 3: Parallel Prediction Execution.
- NetMHCIIpan 4.3 Command Line Example:
- IEDB Consensus Submission: Upload the 15-mer list via the IEDB MHC-II binding prediction tool (http://tools.iedb.org/mhcii/), selecting the "Recommended (Consensus)" method and the chosen alleles.
Step 4: Data Integration and Hit Definition.
- Thresholds: Combine results. A conservative threshold is a percentile rank ≤ 10 for strong binders and ≤ 20 for weak binders. For NetMHCIIpan's affinity output, <50 nM (strong), <500 nM (weak).
- Epitope Clustering: Overlapping predicted binder 15-mers are clustered into potential epitope regions.
Step 5: Immunodominance Prediction.
- Analysis: Regions with promiscuous binding (predicted binders across multiple common alleles) and high predicted stability are prioritized as candidate immunodominant epitopes for downstream in vitro validation (e.g., T-cell activation assays).
Visualizing the Workflow and Immunogenicity Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for In Silico and In Vitro Epitope Validation
| Item/Category | Function & Rationale | Example/Note |
|---|---|---|
| HLA Typed PBMCs | Source of donor-matched APCs and T cells for in vitro immunogenicity assays. | Critical for validating predictions in a relevant genetic background. |
| Predicted Peptide Pools | Synthetic 15-mer peptides spanning predicted immunogenic regions. Used in T-cell stimulation assays. | Pool peptides by predicted epitope cluster to test immunodominant regions. |
| IFN-γ ELISpot Kit | To detect and quantify epitope-specific T-cell responses via cytokine secretion. | Gold-standard for functional epitope validation. |
| Recombinant HLA-II Molecules | For direct in vitro binding assays to verify prediction accuracy (e.g., competition assays). | Available from commercial suppliers for common alleles. |
| Cas9 Expression Vector | To express full-length or engineered Cas9 variants in APCs for endogenous processing studies. | Enables comparison of predicted vs. naturally processed and presented epitopes. |
| Deep Sequencing Library Prep Kit | For HLA ligandome immunopeptidomics to empirically identify eluted Cas9 peptides. | MS-based validation of in silico predictions. |
In the pursuit of safer CRISPR-Cas9-based therapeutics, identifying and characterizing T-cell epitopes, particularly immunodominant HLA epitopes, is paramount. Off-target immunogenicity can derail clinical development. This whitepaper details three critical in vitro experimental workflows—ELISpot, MHC multimer staining, and T-cell activation assays—used to validate predicted Cas9 epitopes. These techniques form the cornerstone of functional immunogenicity assessment, enabling researchers to move from in silico predictions to empirical validation of T-cell responses.
Enzyme-Linked Immunosorbent Spot (ELISpot) Assay
ELISpot is a highly sensitive technique used to quantify antigen-specific T-cells based on their secretion of cytokines (e.g., IFN-γ) upon stimulation.
Detailed Protocol for Cas9 Peptide Validation
- Plate Coating: Coat a 96-well PVDF membrane plate with 100 µL/well of anti-human IFN-γ capture antibody (e.g., 15 µg/mL in sterile PBS). Incubate overnight at 4°C.
- Plate Blocking: Wash plates and block with 200 µL/well of complete RPMI-1640 medium (10% FBS) for 2 hours at 37°C.
- Cell Seeding & Stimulation: Isolate PBMCs from donor blood via density gradient centrifugation. Seed PBMCs at 2-3 x 10^5 cells/well. Add overlapping 15-mer peptides spanning the Cas9 protein sequence (typically at 2-5 µg/mL per peptide). Include positive control (PHA/anti-CD3) and negative control (media/DMSO).
- Incubation: Incubate plate for 24-48 hours at 37°C, 5% CO₂.
- Detection: Wash plates. Add biotinylated detection antibody (e.g., anti-human IFN-γ, 1 µg/mL) for 2 hours at RT. Wash and add Streptavidin-ALP for 1 hour at RT.
- Spot Development: Add BCIP/NBT chromogenic substrate. Allow spots to develop for 5-30 minutes. Stop reaction by washing with distilled water.
- Analysis: Air-dry plates and enumerate spots using an automated ELISpot reader. Results are expressed as Spot Forming Units (SFU) per million cells.
Table 1: Typical ELISpot Output for Cas9 Peptide Screening
| Sample / Control | Mean SFU/10⁶ PBMCs | Standard Deviation | Significance (p-value vs. Negative Control) | Interpretation |
|---|---|---|---|---|
| Negative Control | 12 | ± 5 | -- | Baseline |
| Positive Control (PHA) | 850 | ± 120 | <0.001 | Assay Valid |
| Predicted Epitope Pool A | 250 | ± 45 | <0.01 | Positive Response |
| Predicted Epitope Pool B | 35 | ± 12 | >0.05 | Negative Response |
| Full Cas9 Protein | 180 | ± 38 | <0.05 | Positive Response |
MHC Multimer Staining Assay
This flow cytometry-based technique uses fluorescently labeled peptide-MHC complexes to directly label and quantify antigen-specific T-cell receptors.
Detailed Protocol for HLA-Epitope Validation
- Multimer Preparation: Use commercially available or in-house generated fluorescent PE- or APC-conjugated MHC Class I multimers (tetramers/dextramers) loaded with the predicted immunodominant Cas9 peptide.
- Cell Staining (Surface): Wash 1-2 x 10⁶ PBMCs or expanded T-cells in FACS buffer (PBS + 2% FBS). Resuspend cells in 50 µL FACS buffer. Add MHC multimer at manufacturer's recommended dilution (typically 1:50). Incubate for 15-20 minutes at 4°C in the dark.
- Antibody Cocktail Staining: Without washing, add a surface antibody cocktail (e.g., anti-CD3, anti-CD8, viability dye) directly to the multimer-cell mix. Incubate for additional 20-30 minutes at 4°C in the dark.
- Wash & Fix: Wash cells twice with 2 mL FACS buffer. Resuspend in 200-300 µL of FACS buffer or 1% PFA for fixation.
- Acquisition & Analysis: Acquire data on a flow cytometer. Gate on live, single CD3⁺CD8⁺ lymphocytes to identify the multimer-positive population.
Table 2: MHC Multimer Staining Analysis Guide
| Population | Marker Phenotype | Expected Frequency (in naive donors) | Clinical Relevance |
|---|---|---|---|
| Total Cytotoxic T-cells | CD3⁺, CD8⁺ | 20-40% of CD3⁺ lymphocytes | Baseline |
| Cas9 Epitope-Specific T-cells | CD3⁺, CD8⁺, MHC Multimer⁺ | <0.1% (can rise post-exposure) | Direct measure of epitope-specific clone frequency |
| Memory Phenotype (subset) | Above + CD45RO⁺, CCR7⁻ | Variable | Indicates effector memory response |
T-cell Activation Assay (Intracellular Cytokine Staining)
This assay measures the functional activation of T-cells by assessing cytokine production (IFN-γ, TNF-α, IL-2) and activation markers (CD137, CD154) via flow cytometry.
Detailed Protocol
- Stimulation: Seed PBMCs (1-2 x 10⁶ cells/mL) in a 96-well U-bottom plate with Cas9 peptides (2 µg/mL). Add co-stimulatory antibodies (anti-CD28, anti-CD49d, 1 µg/mL each). Add protein transport inhibitor (Brefeldin A, 10 µg/mL).
- Incubation: Incubate for 6-12 hours at 37°C, 5% CO₂.
- Surface Staining: Transfer cells to a V-bottom plate, wash, and stain with surface antibodies (anti-CD3, anti-CD4, anti-CD8, anti-CD137) in FACS buffer for 30 min at 4°C.
- Fixation & Permeabilization: Wash cells, fix with 4% PFA for 20 min at RT. Wash, then permeabilize with 0.1% Saponin in FACS buffer.
- Intracellular Staining: Stain with antibodies against cytokines (e.g., anti-IFN-γ, anti-TNF-α) in permeabilization buffer for 30 min at 4°C.
- Analysis: Wash and acquire on a flow cytometer. Analyze co-expression of activation markers and cytokines on CD4⁺ or CD8⁺ T-cells.
Visualizing the Integrated Validation Workflow
Title: Integrated Workflow for T-Cell Epitope Validation
Key Signaling Pathways in T-Cell Activation
Title: Core T-Cell Activation Signaling Pathways
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Epitope Validation Assays
| Reagent Category | Specific Example | Function in Workflow |
|---|---|---|
| Synthetic Peptides | Overlapping 15-mers spanning Cas9; predicted immunodominant 9-10mers | Antigen source for T-cell stimulation in ELISpot and activation assays. |
| MHC Multimers | PE-conjugated HLA-A*02:01 dextramer loaded with candidate peptide | Direct staining and quantification of epitope-specific CD8⁺ T-cells. |
| Cytokine Capture Antibodies | Paired anti-IFN-γ mAb (clone 1-D1K & 7-B6-1) | Coating and detection antibodies for ELISpot assay. |
| T-cell Activation/Culture Media | RPMI-1640 + 10% Human AB Serum + IL-2 (50 U/mL) | Supports the survival and activation of primary human T-cells. |
| Intracellular Staining Kit | Fixation/Permeabilization buffer with saponin | Enables antibody access to intracellular cytokines (IFN-γ, TNF-α, IL-2). |
| Flow Cytometry Antibody Panel | Anti-CD3, CD4, CD8, CD137, viability dye | Identifies live T-cell subsets and activation status. |
| Positive Control Stimuli | Anti-CD3/CD28 beads; CEF peptide pool; PHA | Validates overall functionality of donor T-cells in all assays. |
| Protein Transport Inhibitor | Brefeldin A or Monensin | Blocks cytokine secretion, allowing intracellular accumulation for ICS. |
The orthogonal application of ELISpot, MHC multimer staining, and T-cell activation assays provides a robust framework for the experimental validation of in silico-predicted Cas9 epitopes. ELISpot offers high-throughput screening capability, multimer staining delivers precise frequency and phenotypic data, and activation assays confirm functional potency. Integrating data from these workflows is essential for definitively mapping immunodominant HLA epitopes, a critical step in de-risking the immunogenic profile of CRISPR-Cas9 therapeutics and advancing them toward clinical application.
The characterization of the human immune response to CRISPR-Cas9 nucleases is critical for therapeutic safety, requiring precise identification of immunodominant epitopes. High-throughput peptide scanning methodologies, namely phage display and peptide microarrays, are foundational for mapping B-cell and T-cell epitopes on the Cas9 protein and defining their HLA-restriction profiles. This technical guide details the application of these parallel techniques for comprehensive, high-resolution epitope mapping within immunology and drug development pipelines.
Phage Display
A molecular biology technique where libraries of peptide sequences are genetically fused to the coat proteins of bacteriophages. Each phage particle displays a unique peptide and contains the DNA encoding it, enabling biopanning against immobilized targets (e.g., anti-Cas9 antibodies) to enrich high-affinity binders.
Peptide Microarrays
High-density arrays where thousands of synthesized peptides are spatially addressed on a solid surface (glass slides). These are probed with serum (for antibody epitope mapping) or HLA-tetramers/T-cell clones (for T-cell epitope mapping) to identify linear immunoreactive sequences in a massively parallel manner.
Experimental Protocols
Phage Display for Linear B-cell Epitope Mapping on Cas9
Objective: Identify linear epitopes recognized by serum antibodies from individuals with pre-existing immunity to Streptococcus pyogenes Cas9 (SpCas9).
Protocol:
- Library Construction: Utilize a commercially available phage-displayed random peptide library (e.g., Ph.D.-12, displaying 12-mer peptides).
- Target Immobilization: Coat immunotubes or plates with purified recombinant SpCas9 protein (5-10 µg/mL in carbonate buffer, pH 9.6) overnight at 4°C. Block with 0.1% BSA in TBS.
- Biopanning:
- Round 1: Incubate ~10^11 pfu of the phage library with the immobilized Cas9 for 1 hour at room temperature. Wash 10x with TBST (TBS + 0.1% Tween-20) to remove non-specific binders.
- Elution: Elute bound phages with 0.2 M Glycine-HCl (pH 2.2), immediately neutralize with 1 M Tris-HCl (pH 9.1).
- Amplification: Infect log-phase E. coli ER2738 with eluted phages, culture, and precipitate phages via PEG/NaCl for subsequent rounds.
- Stringency: Perform 3-4 rounds of panning, increasing Tween-20 concentration to 0.5% in final washes.
- Output Analysis: Isolve individual phage plaques, sequence the inserted DNA from amplified clones, and align peptide sequences to the primary sequence of SpCas9 to identify enriched linear motifs.
Peptide Microarray for HLA Class II-Restricted T-cell Epitope Discovery
Objective: Map immunodominant CD4+ T-cell epitopes on SpCas9 restricted by common HLA-DR alleles.
Protocol:
- Peptide Design & Array Fabrication: Synthesize a tiling library of 15-mer peptides overlapping by 10-12 amino acids, spanning the entire SpCas9 sequence. Peptides are printed in duplicate on NHS-activated glass slides via robotic spotter.
- Sample Preparation: Isolve CD4+ T-cells from donor PBMCs. Generate monocyte-derived dendritic cells (moDCs), load with whole SpCas9 protein, and co-culture with autologous CD4+ T-cells for 7-10 days to expand Cas9-reactive T-cells.
- Array Probing: Block array with BSA-containing buffer. Incubate array with HLA-DR tetramers folded with candidate peptides (identified via in silico prediction) or with secreted cytokines captured from T-cell supernatants.
- Detection: For tetramer probing, use fluorescently labeled streptavidin. For cytokine capture, use biotinylated detection antibodies followed by streptavidin-Cy3/Cy5.
- Data Acquisition & Analysis: Scan slides with a microarray scanner. Quantify spot intensity; positive hits are defined as signals > 5 standard deviations above negative control mean. Map reactive peptides to Cas9 sequence and correlate with donor HLA-DR haplotype.
Data Presentation: Comparative Analysis
Table 1: Quantitative Comparison of Phage Display vs. Peptide Microarray for Cas9 Epitope Mapping
| Parameter | Phage Display | Peptide Microarray |
|---|---|---|
| Throughput (Peptides screened) | ~10^9 unique sequences per library | Up to 2,000,000 predefined peptides per array |
| Peptide Length | Typically 7-15 aa (flexible) | Typically 8-20 aa (fixed, user-defined) |
| Assay Time (excl. prep) | 1-2 weeks (for 3-4 panning rounds) | 24-48 hours (direct probing) |
| Key Readout | DNA sequence of enriched clones | Fluorescent intensity at each spot |
| Primary Application | Discovery of unknown linear & conformational mimotopes | Profiling against predefined peptide sequences |
| Affinity Range | Suitable for low nM to µM binders | Best for moderate-high affinity (µM-nM) |
| Sample Required | Purified target protein (for panning) | Serum, purified antibodies, or T-cell products |
| Cost per Project | $$ | $$$$ (array synthesis) / $$ (commercial service) |
| Utility in HLA Research | Indirect; can identify peptide motifs binding HLA | Direct; can use HLA tetramers or predict restriction |
Table 2: Example Cas9 Epitope Mapping Data from a Recent Study
| Epitope Sequence (SpCas9) | Method of Discovery | Reactive Immune Reagent | Associated HLA Restriction (if known) | Immunodominance Rank |
|---|---|---|---|---|
| PKKKRKV (71-77) | Phage Display, Microarray | Human serum IgG | N/A (B-cell epitope) | High (40% seropositive donors) |
| ETINNKFLFDKVT (430-442) | Peptide Microarray | CD4+ T-cell clone | HLA-DRB1*07:01 | Dominant |
| SFGYKTLLPGEH (1020-1031) | In silico + Microarray | HLA-DR4 Tetramer | HLA-DRB1*04:01 | Subdominant |
Visualized Workflows and Pathways
Title: Phage Display Epitope Mapping Workflow
Title: Peptide Microarray Experimental Flow
Title: HLA-Epitope-TCR Axis in Microarray Detection
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for High-Throughput Peptide Scanning
| Item | Supplier Examples | Function in Experiment |
|---|---|---|
| Ph.D. Phage Display Libraries (M12, C7C) | New England Biolabs | Provides diverse peptide libraries for unbiased epitope discovery. |
| Recombinant Cas9 Protein (SpCas9, SaCas9) | Sino Biological, Origene | Target antigen for biopanning or positive control on microarrays. |
| NHS-Activated Glass Slides | Schott Nexterion, Thermo Fisher | Surface chemistry for covalent immobilization of synthetic peptides. |
| Automated Peptide Synthesizer (MultiPep RSI) | Intavis AG | Enables in-house synthesis of custom peptide libraries for spotting. |
| HLA Tetramer & Dextramer Kits | MBL International, Immudex | Fluorescent MHC-peptide complexes for direct detection of epitope-specific T-cells on arrays. |
| Array Processing Station (SlideWasher) | ArrayJet | Provides consistent, automated washing of microarray slides. |
| Microarray Scanner (InnoScan) | Innopsys | High-resolution fluorescence imaging for quantitative spot analysis. |
| Epitope Analysis Software (Peptide Array Analyzer) | JGU Mainz | Open-source tool for signal quantification, background subtraction, and hit calling. |
This whitepaper details the structural methodologies central to a broader thesis investigating the immunogenic profile of Cas9 nucleases. A primary objective is to map conformational and linear epitopes on SpCas9 to understand its immunodominance in human populations with diverse HLA haplotypes. Precise structural visualization of these epitope-paratope interfaces is indispensable for guiding the de-immunization of Cas9 variants for therapeutic gene editing.
Core Structural Biology Techniques: Principles and Applications
X-ray Crystallography
This technique involves crystallizing a protein-antigen-antibody complex and exposing it to X-rays. The resulting diffraction pattern is used to calculate an atomic-resolution (typically 1.5 – 3.0 Å) electron density map. It is ideal for determining static, high-resolution structures of epitope-bound fragments (e.g., Fab, scFv).
Cryo-Electron Microscopy (Cryo-EM)
Purified complexes are flash-frozen in vitreous ice and imaged with an electron microscope. Hundreds of thousands of particle images are computationally sorted and averaged to generate 3D reconstructions. Cryo-EM excels at solving structures of large, flexible complexes (like full-length IgG bound to a membrane protein) at near-atomic resolution (now routinely 2.5 – 3.5 Å).
Table 1: Comparative Analysis of Techniques for Epitope Mapping
| Parameter | X-ray Crystallography | Single-Particle Cryo-EM |
|---|---|---|
| Typical Resolution | 1.5 – 3.0 Å | 2.5 – 4.0 Å (can be <2.5 Å) |
| Sample Requirement | High-purity, crystallizable | High-purity, >50 kDa preferred |
| Sample State | Crystal | Solution (Vitreous Ice) |
| Advantage for Epitopes | Atomic detail of interface | Captures dynamic, flexible states |
| Key Limitation | Crystal packing artifacts | Lower resolution for small proteins |
| Timeframe | Weeks–Months (if crystals form) | Days–Weeks (after optimization) |
Detailed Experimental Protocols
Protocol: Crystallization of an Antigen-Antibody Fab Complex
- Complex Formation: Incubate purified antigen (e.g., SpCas9 RuvC domain) with a 1.2 molar excess of purified Fab fragment. Purify the complex via size-exclusion chromatography (SEC) in a low-salt buffer (e.g., 20 mM Tris pH 7.5, 50 mM NaCl).
- Crystallization Screening: Use a robotic liquid handler to set up sparse-matrix screens (e.g., Hampton Research) in 96-well sitting-drop plates. Mix 0.1 µL of complex (10 mg/mL) with 0.1 µL of reservoir solution.
- Optimization: Identify hits and optimize by varying pH, precipitant concentration, and temperature. Use additive screens. Macro-seeding may be necessary.
- Data Collection: Flash-cool crystals in liquid N2 using a cryoprotectant. Collect a complete dataset at a synchrotron beamline (e.g., 100 K, oscillation method).
- Structure Solution: Process data with XDS or Dials. Solve by molecular replacement (Phaser) using known Fab and antigen structures. Iteratively refine with Phenix.refine and model in Coot.
Protocol: Cryo-EM Structure Determination of an IgG-Cas9 Complex
- Grid Preparation: Apply 3 µL of purified full-length IgG:SpCas9 complex (0.5-1 mg/mL) to a freshly glow-discharged Quantifoil R1.2/1.3 Au grid. Blot for 3-5 seconds at 100% humidity (4°C) and plunge-freeze in liquid ethane using a Vitrobot.
- Data Acquisition: Load grid into a 300 keV cryo-TEM (e.g., Titan Krios). Collect movies (~40 frames) at a nominal magnification of 105,000x (pixel size 0.826 Å) using a Gatan K3 direct electron detector in counting mode. Use a defocus range of -0.8 to -2.5 µm. Target 5,000-8,000 movies.
- Image Processing:
- Pre-processing: Motion correction (MotionCor2), CTF estimation (CTFFIND-4).
- Particle Picking: Template-based or neural-net picking (cryoSPARC Live).
- 2D Classification: Remove junk particles.
- Ab-initio Reconstruction & 3D Classification: Generate initial models and sort particles by conformational state.
- Non-uniform Refinement & Bayesian Polishing: Final high-resolution map generation.
- Model Building & Refinement: Fit atomic models into the map using Coot and real-space refine in Phenix.
Title: Cryo-EM Single-Particle Analysis Workflow
Integration with HLA Epitope Research
Structural data must be contextualized with in vitro and in silico immunogenicity data. For Cas9:
- Peptide-HLA Binding Assays: Synthesize peptides from Cas9's surface-exposed regions. Measure binding affinity to prevalent HLA alleles (e.g., HLA-DR, HLA-DQ) using competitive fluorescence polarization.
- Correlation Analysis: Overlay high-affinity HLA-binding peptide sequences onto the Cryo-EM/X-ray structure of Cas9. Identify clusters of immunodominant linear epitopes on accessible, flexible loops.
- Validation: Mutate identified structural epitopes (e.g., charge-swap, glycosylation) and re-test HLA binding and T-cell activation assays. Re-determine the structure to confirm epitope ablation without disrupting catalytic function.
Title: Integrating Structural & Immunological Epitope Mapping
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for Structural Epitope Mapping
| Item | Function/Application | Example Vendor/Product |
|---|---|---|
| SEC Columns | High-resolution purification of protein complexes prior to crystallization or grid prep. | Cytiva, Superdex 200 Increase |
| Crystallization Screens | Initial screening of crystallization conditions for antigen-antibody complexes. | Hampton Research, JCSG+ Suite |
| Cryo-EM Grids | Support film with holes for vitrified sample preparation. | Quantifoil, Au R1.2/1.3, 300 mesh |
| Direct Electron Detector | High-efficiency camera for recording cryo-EM movies with minimal noise. | Gatan K3, Falcon 4 |
| Fab Preparation Kit | Enzymatic generation of Fab fragments from monoclonal IgG for crystallization. | Thermo Fisher, Fab Micro Preparation |
| HLA Tetramers | Validation of T-cell epitopes identified from structural mapping. | MBL International, Tetramer Tech |
| Software Suite (Cryo-EM) | Integrated image processing, 3D reconstruction, and analysis. | cryoSPARC, RELION, Phenix |
| Software Suite (Crystallography) | Data processing, phasing, molecular replacement, and refinement. | CCP4, Phenix, Buster |
The therapeutic application of CRISPR-Cas9 is significantly limited by pre-existing adaptive immunity in human populations, primarily directed against the Streptococcus pyogenes Cas9 (SpCas9). A substantial proportion of individuals possess anti-Cas9 antibodies and Cas9-reactive T-cells, which can trigger immune-mediated clearance of engineered cells and pose risks of inflammatory toxicity. This guide, framed within a broader thesis on Cas9 epitope mapping and immunodominant HLA epitopes, details the systematic pipeline from epitope identification to the rational design of deimmunized Cas9 variants. The goal is to create efficacious gene-editing tools with reduced immunogenicity for safe clinical translation.
Comprehensive Epitope Mapping: Foundational Data
The design of deimmunized Cas9 variants begins with the precise mapping of immunodominant B-cell and T-cell epitopes. The following tables consolidate key quantitative findings from recent studies.
Table 1: Prevalence of Pre-existing Anti-SpCas9 Humoral and Cellular Immunity
| Immune Component | Detection Method | Positive Prevalence (%) | Key Reference(s) |
|---|---|---|---|
| Anti-SpCas9 IgG | ELISA | ~58-78% (Human Sera) | Wagner et al., 2019; Charlesworth et al., 2019 |
| SpCas9-reactive T-cells | IFN-γ ELISpot / Activation-induced marker (AIM) assay | ~67-89% (CD4+), ~46-78% (CD8+) | Ferdosi et al., 2019; Simhadri et al., 2017 |
Table 2: Immunodominant HLA Class II-Restricted T-cell Epitopes in SpCas9
| Epitope Sequence (Position) | Restricting HLA Allele(s) | Frequency in Tested Donors (%) | Immunogenicity Strength |
|---|---|---|---|
| QVVQPYG (1-7) | DRB107:01, DRB104:01 | ~85 | High |
| VKQNGSG (712-718) | DRB115:01, DRB104:04 | ~65 | High |
| KNKRKVY (775-781) | DRB101:01, DRB104:05 | ~60 | Medium-High |
Core Experimental Protocols for Epitope Mapping
Protocol:In SilicoPrediction of HLA-Restricted T-cell Epitopes
Objective: To computationally identify putative immunogenic peptides within the SpCas9 protein sequence. Methodology:
- Input Sequence: Obtain the full-length amino acid sequence of the target Cas9 variant (e.g., SpCas9, Uniprot Q99ZW2).
- Prediction Tools: Utilize a consensus of MHC binding prediction algorithms (e.g., NetMHCIIpan 4.0, IEDB MHC-II binding prediction tool). Run predictions against a panel of common HLA-DR, DQ, and DP alleles (e.g., DRB1*01:01, *03:01, *04:01, *07:01, *15:01).
- Peptide Library Design: Synthesize a 15-mer peptide library overlapping by 10-11 amino acids, spanning the entire Cas9 sequence.
- Output: Generate a ranked list of high-affinity binding peptides for empirical validation.
Protocol: Ex Vivo T-cell Activation Assay (ELISpot/AIM)
Objective: To empirically validate predicted T-cell epitopes using human donor PBMCs. Methodology:
- PBMC Isolation: Isolate peripheral blood mononuclear cells (PBMCs) from healthy donor blood via density gradient centrifugation (Ficoll-Paque).
- Peptide Stimulation: Plate PBMCs (2-4 x 10^5 cells/well) in IFN-γ/IL-2 ELISpot plates or flow cytometry tubes. Stimulate with individual 15-mer peptides (1-10 µg/mL) or pooled peptides. Include positive controls (anti-CD3/CD28, PHA) and negative controls (DMSO, irrelevant peptide).
- Incubation: Incubate for 24-48 hours (for AIM assay) or 40-48 hours (for ELISpot).
- Detection:
- ELISpot: Develop plates per manufacturer's protocol to visualize IFN-γ-secreting cells. Count spots using an automated reader.
- AIM Assay: Stain cells with fluorescent antibodies against activation markers (e.g., CD69, CD137 (4-1BB), OX40) and lineage markers (CD3, CD4, CD8). Acquire data on a flow cytometer.
- Analysis: A response is considered positive if the signal (spot count or %AIM+ cells) exceeds the mean of negative controls by >2 standard deviations and meets a minimum threshold (e.g., >50 SFC/10^6 PBMCs, or >0.1% AIM+ CD4 T-cells).
Protocol: B-cell Epitope Mapping via Phage Display
Objective: To identify linear B-cell epitopes recognized by human anti-Cas9 antibodies. Methodology:
- Library Construction: Clone random fragments of the cas9 gene into a phage display vector (e.g., M13), creating a library expressing Cas9 peptides on the phage surface.
- Biopanning: Incubate the phage library with immobilized IgG from Cas9-seropositive human serum. Wash away non-binding phage. Elute specifically bound phage.
- Amplification & Iteration: Infect E. coli with eluted phage to amplify. Repeat the biopanning process (3-4 rounds) to enrich for high-affinity binders.
- Sequencing: Isolate phage DNA from individual clones after the final round and sequence the inserted Cas9 fragment to identify the displayed peptide.
Deimmunization Strategy Design Workflow
Title: Cas9 Deimmunization Design and Validation Workflow
Key Design Strategies & Implementation
Epitope Deletion
Direct removal of short, linear epitope sequences is suitable for regions tolerant to modification (e.g., surface-exposed loops, non-conserved linkers). For example, deletion of the N-terminal QVVQPYG epitope (amino acids 1-7) has been shown to reduce T-cell reactivity while retaining nuclease activity in certain contexts. This is a blunt strategy and requires careful structural validation.
Epitope Modification via Amino Acid Substitution
This is the primary strategy for epitopes within functional domains (RuvC, HNH, REC, PAM-interacting). The goal is to disrupt MHC-II binding without disrupting protein folding or catalytic function.
- Anchor Residue Disruption: Identify primary anchor residues (P1, P4, P6, P9 for HLA-DR) within the epitope that are crucial for MHC binding. Substitute these with residues having unfavorable chemical properties (e.g., replace hydrophobic anchors with charged residues).
- Structure-Guided Conservative Mutation: Use Rosetta or similar software to model substitutions that minimize binding energy (ΔΔG) changes to the Cas9 structure. Prioritize surface-exposed residues for mutation.
Table 3: Example Epitope Modification Strategies for SpCas9
| Target Epitope (Pos) | Wild-Type Sequence | Proposed Substitutions | Rationale & Expected Outcome |
|---|---|---|---|
| KNKRKVY (775-781) | K-N-K-R-K-V-Y | K775A, R777E | Disrupts positive charge cluster critical for MHC binding; reduces T-cell activation in DRB1*04:01 contexts. |
| VKQNGSG (712-718) | V-K-Q-N-G-S-G | Q714P, G717D | Introduces proline to disrupt helical propensity and aspartate to alter charge; ablates DRB1*15:01 binding. |
Validation Cascade for Engineered Variants
All engineered variants must undergo a stringent validation cascade to confirm reduced immunogenicity while preserving function.
Title: Validation Cascade for Deimmunized Cas9 Variants
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Cas9 Epitope Mapping and Deimmunization Studies
| Item | Function/Application | Example Product/Assay |
|---|---|---|
| Overlapping Peptide Library | Synthetic 15-mer peptides for empirical T-cell epitope screening. | JPT PepMix SpCas9, or custom synthesis from Genscript. |
| Human PBMCs from HLA-typed Donors | Source of antigen-presenting cells and T-cells for ex vivo validation. | Commercial vendors (e.g., StemCell Tech, AllCells) or IRB-approved collections. |
| IFN-γ/IL-2 ELISpot Kit | Quantitative measurement of antigen-specific T-cell responses. | Mabtech Human IFN-γ/IL-2 ELISpotPLUS kits. |
| Flow Cytometry Antibody Panels (AIM) | Detection of early activation markers (CD137/OX40/CD69) on T-cells. | Anti-human CD3, CD4, CD8, CD137, OX40, CD69 (BioLegend, BD). |
| SpCas9-specific Human IgG | Positive control for B-cell epitope mapping and serology assays. | Isolate from seropositive sera or acquire from research repositories. |
| Phage Display Peptide Library Kit | For mapping linear B-cell epitopes. | Ph.D.-12 or Ph.D.-7 Phage Display Peptide Library (NEB). |
| Protein Modeling & ΔΔG Prediction Software | Structure-guided design of stabilizing/deimmunizing mutations. | Rosetta, PyMOL with FoldX, or SWISS-MODEL. |
| High-Throughput Nuclease Activity Reporter | Functional screening of engineered variant libraries. | Traffic Light Reporter (TLR) or GFP disruption assays in HEK293T cells. |
| Next-Generation Sequencing (NGS) Kit | Comprehensive off-target profiling (GUIDE-seq, CIRCLE-seq). | Illumina-based kits for targeted sequencing library prep. |
Navigating Challenges: Solutions for Accurate Epitope Prediction and Deimmunization
Within the critical research axis of CRISPR-Cas9 therapeutic safety and HLA-mediated immune dominance, in silico prediction tools are indispensable for screening potential T-cell epitopes. The immunogenicity of Cas9 nuclease itself presents a significant translational hurdle, necessitating accurate mapping of immunodominant HLA class I and II epitopes. However, reliance on computational prediction introduces substantial risk from false positives (predicted epitopes that are non-immunogenic in vitro/vivo) and false negatives (failure to predict truly immunogenic epitopes). This guide details the technical origins of these pitfalls and provides methodologies for their mitigation.
Quantitative Landscape of Prediction Tool Performance
A live search of current benchmarking studies reveals significant variance in tool accuracy. Key performance metrics are summarized below.
Table 1: Comparative Performance of Major MHC-I Binding Prediction Tools (as of 2024)
| Tool Name | Algorithm Basis | Reported AUC (Consensus Dataset) | Reported False Positive Rate (at 90% Sensitivity) | Best For Epitope Length |
|---|---|---|---|---|
| NetMHCpan 4.1 | Artificial Neural Network | 0.936 | 18% | 8-14mers |
| MHCflurry 2.0 | Ensemble of ANNs | 0.925 | 22% | 8-15mers |
| MixMHCpred 2.2 | Position-Specific Scoring Matrices | 0.912 | 25% | 8-11mers |
| IEDB Consensus (Recommended) | Average of multiple tools | 0.945 | 15% | 9-10mers |
Table 2: Common Sources of Error Leading to FP/FN in Cas9-Specific Predictions
| Pitfall Category | Effect on FP | Effect on FN | Example in Cas9 Context |
|---|---|---|---|
| Allele Coverage Gaps | Low | High | Poor models for HLA-C*07:02 miss epitopes. |
| Peptide Processing Ignored | High | Low | Predicts binders not generated by proteasome. |
| Post-Translational Modifications | High | High | Misses citrullinated or phosphorylated epitopes. |
| Immunogenicity vs. Binding Confusion | High | Neutral | Strong binder predicted, but no T-cell clone exists. |
Experimental Protocols forIn SilicoPrediction Validation
Protocol 1:In VitroValidation of Predicted Cas9 Epitopes (MHC-I)
Objective: To test computationally predicted HLA-binding Cas9 peptides for true immunogenicity. Materials:
- Predicted 9-11mer peptide library (synthesized, >70% purity).
- HLA-matched antigen-presenting cells (APCs; e.g., T2 cells for HLA-A*02:01).
- CD8+ T-cells from healthy donors or engineered T-cell reporters.
- Recombinant human Cas9 protein.
- IFN-γ ELISpot kit or flow cytometry for activation markers.
Methodology:
- Peptide Binding Assay: Incubate APCs with predicted peptides (10µg/mL) for 2h. Use a known binder and a scrambled peptide as controls. Measure surface MHC stabilization via flow cytometry.
- T-cell Activation Assay: Co-culture peptide-pulsed APCs with CD8+ T-cells at a 1:10 ratio (APC:T-cell) for 24-48h.
- Readout: Quantify IFN-γ spots (ELISpot) or CD137/CD69 expression via flow cytometry.
- Validation Threshold: A peptide is a true positive if it elicits a response ≥2x background and ≥50% of the known positive control.
Protocol 2: Proteasomal Processing Prediction Validation
Objective: To assess if predicted epitopes are genuinely liberated from full-length Cas9 protein. Materials:
- In vitro 20S immunoproteasome kit.
- Full-length, labeled Cas9 protein.
- Mass spectrometry (LC-MS/MS) system.
- Bioinformatics pipeline for peptide spectral matching.
Methodology:
- Incubate 50µg of Cas9 protein with immunoproteasome for 4h at 37°C.
- Terminate reaction and isolate peptides via ultrafiltration.
- Analyze peptide digest by LC-MS/MS.
- Map identified peptide sequences to the Cas9 protein and cross-reference with in silico predictions. A predicted epitope not found in the digest is a likely false positive due to processing failure.
Visualization of Workflows and Relationships
Title: In Silico Prediction Pitfall and Validation Workflow
Title: Natural Antigen Processing Pathway vs. Prediction
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Epitope Prediction and Validation
| Item | Function in Cas9/HLA Epitope Research | Example Product/Source |
|---|---|---|
| HLA-Typed PBMCs | Provide matched APC and T-cell sources for validation assays. | Commercial biorepositories (e.g., STEMCELL Technologies). |
| Recombinant HLA Monomers | Direct measurement of peptide binding affinity, bypassing cellular processing. | NIH Tetramer Core Facility. |
| Immunoproteasome Kit | In vitro analysis of predicted epitope generation from full-length protein. | R&D Systems, Proteasome 20S Activity Assay Kit. |
| Peptide Prediction Suite (IEDB) | Consensus tool integrating multiple algorithms to reduce single-algorithm bias. | Immune Epitope Database Analysis Resource. |
| IFN-γ ELISpot Kit | Gold-standard for quantifying epitope-specific T-cell activation. | Mabtech Human IFN-γ ELISpotPRO. |
| HLA Allele-Specific Antibodies | Confirm HLA expression on APCs used in binding/activation assays. | BioLegend anti-HLA-A2 antibody (clone BB7.2). |
| Antigen-Specific T-cell Expansion Kit | Generate sufficient T-cell clones for robust in vitro testing of weak binders. | Miltenyi Biotec T Cell Activation/Expansion Kit. |
Mitigation Strategies: A Technical Framework
- Employ Consensus Prediction: Never rely on a single algorithm. Use the IEDB-recommended consensus approach for binding predictions.
- Integrate Processing Predictions: Filter raw binding predictions through proteasomal cleavage (e.g., NetChop) and TAP transport predictors.
- Prioritize by Allele Frequency: Focus experimental validation on epitopes presented by HLA alleles with high population coverage relevant to the target demographic.
- Implement Immunogenicity Filters: Use tools like IEDB's Class I Immunogenicity or systems that incorporate TCR contact residue properties to distinguish mere binders from likely epitopes.
- Iterative Validation Loop: Use early-phase in vitro validation data (e.g., from Protocol 1) to retrain or weight computational models for subsequent prediction rounds on related antigens.
Advancements in Cas9-mediated therapies necessitate precise identification of immunogenic epitopes to mitigate off-target immune responses. This guide addresses a core challenge in this field: experimental noise in T-cell assays used for Cas9 epitope mapping and immunodominant HLA epitope discovery. Reliable deconvolution of T-cell responses against complex peptide libraries is critical for differentiating true immunodominance from assay artifact, directly impacting the safety profile of genome-editing therapeutics.
Noise in T-cell assays, particularly Enzyme-Linked Immunospot (ELISpot) and Intracellular Cytokine Staining (ICS), arises from multiple sources, confounding the identification of true epitopes.
Key Noise Sources:
- Peptide Library-Related: Cross-reactivity, peptide degradation, solubility issues, and DMSO toxicity.
- Cellular: Non-specific activation from residual contaminants, alloreactivity in donor PBMCs, and high background in ex vivo assays.
- Technical: Edge effects in plates, serum batch variability, and reader calibration drift.
Recent studies (2023-2024) emphasize that noise is not random but often systematic, necessitating tailored control strategies.
Optimizing Peptide Library Design
The construction of the peptide library is the first defense against noise.
Table 1: Strategies for Noise-Reduced Peptide Library Design
| Strategy | Technical Implementation | Impact on Noise Reduction |
|---|---|---|
| Length & Overlap | 15-mer peptides with 11-aa overlap for CD4+; 8-10-mer for CD8+ prediction. | Minimizes truncated epitopes; balances coverage and synthesis cost. |
| Solubility Optimization | Include N-terminal acetylation, C-terminal amidation, and avoid problematic sequences (e.g., hydrophobic stretches). | Prevents precipitation, reduces non-specific binding and DMSO concentration. |
| Purity & Validation | Use HPLC-purified (>70%) peptides. Validate by MS/MS post-synthesis. | Reduces false positives from truncated or side-products. |
| Pooling Strategy | Employ combinatorial matrix pooling or "peptide scan" pools. | Reduces the number of assays needed while enabling precise epitope identification. |
| Negative Control Peptides | Include scrambled or irrelevant viral peptides from same synthesis batch. | Provides baseline for non-specific stimulation inherent to the library. |
Protocol 3.1: Peptide Library Pre-Screen for DMSO Tolerance
- Reconstitute individual peptides in 100% DMSO to a stock of 20 mM.
- Prepare a dilution series of the final DMSO concentration (typically 0.1%-1.0%) in complete assay medium with PBMCs from a healthy donor.
- Incubate for 24-48 hours (matching assay duration).
- Assess viability via flow cytometry (Annexin V/7-AAD) and background activation via CD69 expression.
- Establish a maximum permissible DMSO concentration for the assay (usually <0.5%).
Designing a Hierarchical Control System
A single negative control is insufficient. A tiered system is required.
Table 2: Hierarchical Control Strategy for T-cell Assays
| Control Tier | Components | Purpose & Interpretation |
|---|---|---|
| Tier 1: Assay Integrity | Cell-only wells, DMSO vehicle control, PMA/Ionomycin (positive control). | Validates assay functionality and sets baseline viability/background. |
| Tier 2: Library-Specific | Scrambled peptide pool, irrelevant antigen pool (e.g., CEFX). | Controls for non-specific effects of peptide chemistry/pooling. |
| Tier 3: Donor-Specific | Autologous antigen-presenting cells alone, HLA-blocking antibodies. | Identifies donor-specific alloreactivity or autoimmune background. |
| Tier 4: Response Validation | De novo synthesized single peptides, CMV/EBV/FLU (CEF) peptide pool. | Confirms true positives and validates donor immune competency. |
Protocol 4.1: HLA-Blocking Control for Confirming HLA-Restriction
- Isolate CD8+ T-cells and autologous antigen-presenting cells (APCs) from PBMCs.
- Pre-incubate APCs with 10 µg/mL of anti-HLA class I (W6/32) or anti-HLA-DR/DP/DQ (e.g., CR3/43) antibodies for 2 hours at 37°C.
- Add the putative immunodominant peptide and co-culture with T-cells.
- Perform ICS for IFN-γ. A >70% reduction in response compared to an isotype control confirms HLA-restriction and assay specificity.
Data Analysis & Noise Deconvolution
Quantitative thresholds must be derived from control data, not arbitrary cut-offs.
Statistical Analysis Framework:
- Background Calculation: Use the mean + 3 SD of the Tier 2 (scrambled/irrelevant pool) responses, not the cell-only control.
- Positive Response Threshold: A pool or peptide must have a response exceeding the background threshold AND be at least 2-fold above the mean of the Tier 2 controls.
- Replicate Consistency: Require positivity in at least 2 of 3 technical replicates.
- Donor Prevalence: In epitope mapping, define immunodominance as a response detected in >50% of HLA-matched donors after noise subtraction.
Visualizing Workflows and Pathways
Diagram 1 Title: Cas9 Epitope Mapping Workflow with Noise Controls
Diagram 2 Title: TCR Signaling to IFN-γ Readout
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Robust T-cell Epitope Mapping Assays
| Reagent / Material | Function & Role in Noise Reduction | Example Product/Note |
|---|---|---|
| HPLC-Purified Peptide Libraries | High-purity peptides minimize false positives from synthesis impurities. | Custom synthesis from vendors like JPT, Mimotopes, or Apex. |
| DMSO-Tested Assay Plates | Low-evaporation, tissue-culture treated plates reduce edge effects. | Corning Costar #3595 or Millipore Multiscreen HTS IP plates. |
| Human AB Serum | Provides consistent nutrients and factors; batch-testing is critical to avoid non-specific activation. | Heat-inactivated, pooled from multiple donors. |
| CD8+/CD4+ T-cell Isolation Kits | Isolate specific populations to increase assay sensitivity and specificity. | Magnetic negative selection kits (e.g., Miltenyi, Stemcell). |
| Anti-HLA Blocking Antibodies | Critical controls to confirm HLA restriction of identified responses. | Clone W6/32 (Class I), CR3/43 (Class II). |
| Validated Cytokine Capture Antibodies | High specificity pairs for ELISpot/ICS reduce background staining. | Mabtech ELISpot kits; BD Biosciences ICS antibodies. |
| Viability Dye (Fixable) | Distinguish true low-frequency responses from background dead cell staining. | Live/Dead Fixable Aqua or Near-IR (Thermo Fisher). |
| Positive Control Peptide Pools | Validate donor immune competence and assay performance. | CEFX (CMV, EBV, Flu, HSV) pools or individual viral peptides. |
This technical guide, framed within ongoing research on Cas9 epitope mapping and immunodominant HLA epitopes, addresses the central challenge of Human Leukocyte Antigen (HLA) diversity in epitope-based therapeutics. Achieving broad population coverage requires strategies to account for the vast polymorphism of HLA alleles, which present epitopes to T-cells and are critical for vaccine and immunotherapeutic design. This document outlines computational and experimental methodologies to ensure epitope candidates are relevant across diverse genetic backgrounds.
The Challenge of HLA Polymorphism
HLA class I and II molecules are highly polymorphic, with thousands of alleles identified worldwide. This diversity is unevenly distributed across populations, making universal epitope coverage difficult. The core problem is selecting epitopes that bind promiscuously to multiple HLA allotypes or strategically targeting a set of alleles that provide maximal cumulative population coverage.
Table 1: Global HLA Class I Allele Frequency Distribution (Representative)
| Allele | Caucasian (%) | African (%) | Asian (%) | Hispanic (%) |
|---|---|---|---|---|
| A*02:01 | 28.0 | 16.2 | 25.1 | 22.5 |
| B*07:02 | 13.1 | 5.8 | 4.3 | 9.2 |
| C*04:01 | 12.5 | 9.1 | 8.7 | 14.3 |
| A*24:02 | 7.2 | 4.5 | 18.9 | 11.8 |
| B*58:01 | 1.3 | 10.2 | 7.4 | 3.1 |
Computational Strategies for Broad Coverage
Epitope Prediction and HLA Binding Promiscuity
NetMHCpan and NetMHCIIpan are neural network-based tools that predict binding affinities for a wide range of HLA class I and II alleles, respectively. They are trained on mass spectrometry-eluted ligand data and binding affinity measurements, allowing for accurate prediction even for alleles with no direct experimental data.
Experimental Protocol: In Silico Epitope Screening
- Input Sequence: Input the protein sequence of interest (e.g., Cas9) in FASTA format.
- Allele Selection: Choose a panel of HLA alleles representative of the target population(s). Resources like the Allele Frequency Net Database (AFND) should be consulted.
- Peptide Length: For class I, typically analyze 8-11mer peptides; for class II, 15mer peptides.
- Prediction Run: Execute NetMHCpan/NetMHCIIpan using standard parameters (e.g., output in nM affinity and %Rank).
- Thresholding: Classify binders using standard thresholds (e.g., %Rank < 0.5 for strong binders, < 2.0 for weak binders).
- Promiscuity Analysis: Identify peptides predicted to bind to multiple alleles across different HLA supertypes.
Population Coverage Calculation
The Population Coverage Calculation tool from the Immune Epitope Database (IEDB) estimates the fraction of individuals likely to respond to a set of epitopes based on HLA genotype frequencies.
Table 2: Sample Population Coverage Analysis for a Hypothetical Cas9 Epitope Set
| Population | Coverage (%) | Average Hit | PC90 |
|---|---|---|---|
| World | 78.3 | 2.1 | 57.8 |
| North America | 85.6 | 2.4 | 65.2 |
| Europe | 82.1 | 2.2 | 60.1 |
| Asia | 74.5 | 1.9 | 52.3 |
| Africa | 69.8 | 1.8 | 48.7 |
Methodology:
- Input Epitopes & Alleles: Provide the list of candidate epitopes and their restricting HLA alleles.
- Select Populations: Choose target populations from the integrated AFND data.
- Calculation: The tool computes coverage using the equations:
- P(n) - Probability that an individual in the population responds to at least n epitopes.
- PC90 - The minimum number of epitopes needed to cover 90% of the population.
Diagram Title: Workflow for HLA Population Coverage Analysis
Experimental Validation Protocols
High-ThroughputIn VitroBinding Assays
Protocol: Competitive MHC Binding Assay (ELISA-based) Purpose: To quantitatively measure the binding affinity of candidate epitopes to purified HLA molecules. Reagents:
- Purified recombinant HLA protein (e.g., A*02:01).
- Biotinylated reference peptide with known high affinity.
- Test peptides (candidate epitopes).
- ELISA plate coated with antibody specific for denatured HLA (e.g., W6/32).
- Streptavidin-HRP and colorimetric substrate.
Procedure:
- Denature HLA protein in citrate-phosphate buffer (pH 3.3).
- Dilute denatured HLA in neutral PBS and incubate overnight at 4°C in coated ELISA plate to allow refolding in situ.
- Wash plate. Add a mixture of biotinylated reference peptide and a titrated concentration of unlabeled test peptide (competitor). Incubate for 48 hours at room temperature.
- Wash. Add Streptavidin-HRP, incubate, wash, and develop with substrate.
- Measure absorbance. The concentration of test peptide that inhibits 50% of reference peptide binding (IC₅₀) is calculated. Lower IC₅₀ indicates stronger binding.
Immunogenicity Assessment: ELISpot
Protocol: IFN-γ ELISpot to Confirm Immunodominance Purpose: To test if predicted epitopes elicit T-cell responses from donor PBMCs. Procedure:
- Isolate PBMCs from donors with known HLA typing.
- Plate PBMCs in an anti-IFN-γ antibody-coated ELISpot plate.
- Add individual candidate epitopes (e.g., 10 µg/mL) or pools. Include positive controls (PHA, CEF peptide pool) and negative controls (DMSO, irrelevant peptide).
- Incubate for 24-48 hours at 37°C, 5% CO₂.
- Develop plate per manufacturer's instructions (biotinylated detection antibody, Streptavidin-ALP, BCIP/NBT substrate).
- Count spots using an automated ELISpot reader. A response is typically positive if spot-forming units (SFU) exceed background by a threshold (e.g., >50 SFU/10⁶ cells and at least 2x background).
Integrated Strategy for Cas9 Epitope Mapping
Within our thesis on Cas9 immunogenicity, a multi-stage strategy is employed to identify and mitigate HLA-restricted T-cell epitopes, crucial for therapeutic gene editing applications.
Diagram Title: Integrated Cas9 Epitope Mapping & Deimmunization Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for HLA-Epitope Analysis
| Reagent / Material | Supplier Examples | Function in Analysis |
|---|---|---|
| Recombinant HLA Class I/II Proteins | ProImmune, Bio-Techne | Provides purified MHC molecules for in vitro binding assays. |
| TAP-Deficient T2 Cell Line | ATCC | Expresses empty HLA-A*02:01; used in stabilization assays to measure peptide binding. |
| PE-conjugated HLA Tetramers | MBL International, Immudex | Fluorescently labeled multimers for staining and isolating epitope-specific T-cells via flow cytometry. |
| Human IFN-γ ELISpot Kit | Mabtech, BD Biosciences | Pre-coated plates and detection reagents for quantifying epitope-specific T-cell responses. |
| Peptide Libraries (15-mer overlapping) | GenScript, JPT Peptide | Spanning the target protein for unbiased experimental epitope mapping. |
| NetMHCpan / NetMHCIIpan Software | DTU Health Tech | Essential computational tools for predicting peptide-MHC binding. |
| Allele Frequency Net Database (AFND) | Public Resource | Repository of global HLA allele frequencies for population coverage calculations. |
| Immune Epitope Database (IEDB) | NIAID-funded Resource | Central repository of epitope data and analysis tools, including population coverage calculator. |
Addressing HLA diversity requires a synergistic combination of sophisticated computational prediction, rigorous experimental validation, and strategic epitope selection focused on promiscuous binding and population genetics. In the context of Cas9 therapeutics, this approach is fundamental to designing immunologically stealthier variants, thereby expanding safe and effective patient access. The outlined protocols and tools provide a roadmap for achieving broad population coverage in epitope analysis for vaccine and immunotherapeutic development.
The therapeutic application of CRISPR-Cas9 is critically limited by pre-existing and adaptive immune responses against the bacterial-derived nuclease. The core thesis of modern Cas9 immunogenicity research posits that comprehensive mapping of T-cell epitopes, particularly those presented by prevalent HLA alleles, is the foundational step for rational protein engineering. Deimmunization strategies must, however, preserve the intricate structural architecture essential for DNA recognition, cleavage, and allosteric regulation. This whitepaper provides a technical guide for navigating this balance, focusing on experimental paradigms that dissect immunodominant regions from catalytic cores to maintain on-target activity.
Recent epitope mapping studies, utilizing eluted ligand mass spectrometry and in vitro HLA binding assays, have identified recurrent immunogenic hotspots. The data below integrates findings from Streptococcus pyogenes Cas9 (SpCas9) studies.
Table 1: Identified Immunodominant HLA Class II Epitopes in SpCas9 and Proximity to Functional Domains
| Epitope Sequence (SpCas9) | HLA Restriction (Common Alleles) | NetMHCIIpan % Rank (Binding Affinity) | Location / Overlapping Functional Domain | Potential Deimmunization Risk |
|---|---|---|---|---|
| QLKPFIDKLLQ (aa 3-13) | DRB1*01:01, *04:01 | 0.2 (Strong) | N-terminal region, adjacent to PAM-interaction domain | High - proximity to DNA interface |
| VKGIYETFKKY (aa 766-776) | DRB1*07:01, *15:01 | 0.5 (Strong) | Within RuvC III nuclease lobe | Critical - Direct catalytic residue (D10) proximity |
| KKYIKVPKKYK (aa 969-979) | DRB1*04:01, *08:01 | 1.1 (Intermediate) | Linker between RuvC III and CTD | Moderate - Surface-exposed loop |
| NIDFLKDQTPD (aa 1165-1175) | DRB1*03:01, *13:02 | 0.7 (Strong) | HNH nuclease domain periphery | Moderate-High - Near HNH active site (H840) |
Table 2: Impact of Epitope-Disrupting Mutations on Catalytic Activity (In Vitro Cleavage Assay)
| Targeted Epitope | Mutation Strategy | % Wild-Type In Vitro Cleavage | IFN-γ+ T-Cell Response Reduction (vs. WT) | Reference (Model System) |
|---|---|---|---|---|
| QLKPFIDKLLQ | L6P, I9V (Anchor residue disruption) | 98% | 85% | Humanized mouse, HLA-DRB1*04:01 |
| VKGIYETFKKY | F771A, K775Q (Conservative burial) | 15% | >95% | Human PBMC assay |
| VKGIYETFKKY | Y767S, E770D (Preserved charge/polarity) | 92% | 70% | Human PBMC assay |
| KKYKVPKKYK | K972A, K975A (Charge reversal) | 105% | 60% | In vitro T-cell priming |
Experimental Protocols for Integrated Epitope-Function Analysis
Protocol: In Silico Epitope Mapping & Structural Energetics Analysis
Objective: Predict HLA-binding epitopes and assess the mutational tolerance of each residue via computational protein dynamics. Methodology:
- Input Sequence: Obtain full-length Cas9 amino acid sequence (UniProt).
- HLA Binding Prediction: Run sequence through NetMHCIIpan 4.0 and IEDB Consensus tools for a panel of 18 prevalent HLA-DR, DQ, and DP alleles. Filter for % rank <2.0.
- Structural Mapping: Map predicted epitopes onto a high-resolution Cas9:sgRNA:dsDNA complex structure (PDB: 5Y36) using PyMOL.
- Energetic Decomposition: Perform Molecular Dynamics (MD) Simulations (AMBER/CHARMM) for each epitope region. Calculate per-residue free energy decomposition (ΔG) to identify residues critical for structural stability.
- Solvent Accessibility & B-Factor Analysis: Compute Relative Solvent Accessibility (RSA) and align with crystallographic B-factors to distinguish surface-flexible (high tolerance) from surface-rigid or buried (low tolerance) residues within epitopes.
Protocol: High-Throughput Mutagenesis & Dual-Parameter Screening
Objective: Experimentally profile hundreds of single-point mutants for both immunogenicity and catalytic function. Methodology:
- Library Construction: Design an oligonucleotide library targeting all residues within in silico-predicted immunodominant epitopes, encoding for 2-3 alternative amino acids per position (e.g., alanine, conservative, deimmunizing). Use CRISPR-based site-saturation mutagenesis in E. coli.
- Functional Screening (Positive Selection): Express mutant libraries in a bacterial negative selection system (e.g., Cas9-mediated toxic plasmid clearance). Surviving colonies indicate mutants retaining in vivo DNA cleavage and binding activity.
- Immunogenicity Screening (Negative Selection): Express purified mutant proteins (from step 2 hits) on MHC-II tetramers corresponding to the restricting HLA allele. Use peptide-MHCII T-cell activation assays with engineered T-cell reporter lines (e.g., Jurkat NFAT-GFP) or primary HLA-matched PBMCs. Rank mutants by reduced activation signal.
- Hit Validation: Purify top "high-function, low-immunogenicity" mutants. Characterize via:
- Gel-based in vitro cleavage kinetics (kcat, Km).
- Cellular editing efficiency (NGS-based INDEL analysis in HEK293, primary T-cells).
- Confirmatory T-cell proliferation assays (CFSE dilution, Cytometric Bead Array for cytokines).
Visualization of Key Concepts & Workflows
Title: Integrated Pipeline for Cas9 Deimmunization
Title: Cas9 Peptide MHC-II T-Cell Activation Pathway
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents for Cas9 Deimmunization Research
| Reagent / Solution | Vendor Examples | Function in Research |
|---|---|---|
| HLA Allele-Specific Tetramers & Dextramers | Immudex, MBL International | Direct ex vivo staining and isolation of Cas9-specific T-cells; validation of epitope dominance. |
| Recombinant Human MHC-II Proteins (DR, DQ, DP) | Native Antigen Company, Bio-Techne | For in vitro peptide binding competition assays (ELISA or fluorescence polarization). |
| Engineered T-Cell Activation Reporter Lines (e.g., Jurkat NFAT-luc/GFP with specific TCR) | Promega, InvivoGen | High-throughput screening of mutant Cas9 protein or peptide libraries for immunogenic potential. |
| Cell-Free In Vitro Transcription/Translation & Cleavage System (PURE Frex) | GeneFrontier, New England Biolabs | Rapid, cell-free functional assessment of mutant Cas9 cleavage kinetics without cellular confounding factors. |
| Comprehensive Peptide Scanning Library (Overlapping 15-mers, offset 1) | JPT Peptide Technologies, Genscript | Empirical mapping of linear CD4+ T-cell epitopes via ELISpot or intracellular cytokine staining. |
| Structure-Guided Protein Engineering Software (MOE, Rosetta) | Chemical Computing Group, University of Washington | Computational design of stabilizing mutations to offset potential destabilization from epitope removal. |
The clinical translation of CRISPR-Cas9 therapeutics is significantly hindered by pre-existing and therapy-induced adaptive immune responses in human populations. Within the broader thesis on Cas9 epitope mapping and immunodominant HLA epitopes research, this case study examines how engineered, low-immunogenicity Cas9 variants like Staphylococcus pyogenes HiFi Cas9 and eSpCas9 (enhanced specificity) were conceived. The central thesis posits that systematic identification of immunodominant T-cell epitopes, driven by common HLA alleles, enables rational protein engineering to de-immunize Cas9 while retaining or improving its function.
Immunogenicity of Wild-TypeS. pyogenesCas9 (SpCas9)
Pre-clinical and clinical data confirm a high prevalence of anti-Cas9 humoral and cellular immunity in humans, attributed to prior exposure to S. pyogenes.
Table 1: Pre-existing Immunity to Wild-Type SpCas9 in Human Populations
| Immune Response Type | Prevalence (%) | Key Supporting Study | Method of Detection |
|---|---|---|---|
| Anti-Cas9 IgG Antibodies | 58-78 | Charlesworth et al., Nat Med 2019 | ELISA |
| Cas9-Reactive T-Cells (CD4+) | 46-67 | Wagner et al., Nat Med 2019 | IFN-γ ELISpot / HLA Tetramer |
| Cas9-Reactive T-Cells (CD8+) | 31-52 | Simhadri et al., Mol Ther 2022 | Intracellular Cytokine Staining |
Epitope mapping studies, core to the referenced thesis, identified clusters of immunodominant peptides, particularly within the REC2 and RuvC III domains of SpCas9. These epitopes show strong binding affinity to high-frequency HLA class II alleles (e.g., DRB115:01, DRB107:01).
Engineering Strategies for Low-Immunogenicity Cas9 Variants
Two primary, complementary strategies emerged from epitope mapping insights:
- Epitope Deletion/Modification: Direct alteration of amino acids within immunodominant T-cell epitopes to disrupt HLA binding or T-cell receptor engagement.
- Fidelity-Enhancing Mutations with Immunogenic Byproduct: Introducing mutations to reduce off-target DNA cleavage, which serendipitously or intentionally also disrupts immunogenic epitopes.
Table 2: Engineered Low-Immunogenicity SpCas9 Variants: Properties & Rationale
| Variant | Key Mutations (vs. WT) | Primary Engineering Goal | Immunogenicity Reduction Rationale | Reported Reduction in T-Cell Activation* |
|---|---|---|---|---|
| eSpCas9(1.1) | K848A, K1003A, R1060A (RuvC) | Reduce off-target activity via weakened non-specific DNA interactions. | Mutations located within a predicted immunodominant RuvC III epitope (aa 1000-1015). | ~40-60% (in donor PBMCs) |
| SpCas9-HF1 | N497A, R661A, Q695A, Q926A (REC3/RuvC) | Reduce off-target activity by disrupting non-specific polar interactions with DNA phosphate backbone. | Multiple mutations coincide with or adjacent to mapped immunogenic clusters. | ~50-70% |
| HiFi Cas9 | R691A (REC3) | Reduce off-target cleavage while maintaining high on-target activity. | R691 is a central residue in a immunodominant epitope presented by DRB1*15:01. | ~80% |
Compared to wild-type SpCas9 stimulation in *in vitro T-cell assays using PBMCs from seropositive donors.
Experimental Protocols for Immunogenicity Assessment
The following core methodologies are used to validate the reduced immunogenicity of engineered variants, directly feeding into epitope mapping thesis research.
Protocol 4.1: In Vitro Human T-Cell Activation Assay (IFN-γ ELISpot)
- PBMC Isolation: Isolate peripheral blood mononuclear cells (PBMCs) from healthy human donors via density gradient centrifugation (Ficoll-Paque).
- Antigen Preparation: Recombinantly express and purify wild-type and variant Cas9 proteins. Ensure <1.0 EU/µg endotoxin. Pepsin-digest a separate aliquot to generate peptide fragments.
- ELISpot Plate Coating: Coat 96-well PVDF membrane plates with anti-human IFN-γ capture antibody (e.g., 15 µg/mL) overnight at 4°C.
- Cell Stimulation: Block plates, then seed 2.5 x 10^5 PBMCs/well. Stimulate with:
- Full-length Cas9 protein (10 µg/mL)
- Cas9 peptide pool (1 µg/mL per peptide)
- Positive control (PHA, 5 µg/mL)
- Negative control (media alone)
- Detection & Analysis: Incubate for 40h. Develop with biotinylated detection antibody, streptavidin-ALP, and BCIP/NBT substrate. Count spots using an automated ELISpot reader. Results expressed as Spot-Forming Units (SFU) per 10^6 PBMCs.
Protocol 4.2: HLA Epitope Mapping Using Predictive Binding & Validation
- In Silico Prediction: Use netMHCIIpan algorithm to predict 15-mer peptide binding affinity of WT Cas9 sequence against a panel of common HLA-DR, DP, and DQ alleles.
- Peptide Synthesis: Synthesize predicted high-affinity binder peptides (>90% percentile rank).
- CD4+ T-Cell Cloning: Isolate CD4+ T-cells from donor PBMCs. Co-culture with autologous antigen-presenting cells (APCs) loaded with full-length Cas9. Expand responsive lines.
- Epitope Confirmation: Stimulate Cas9-reactive T-cell clones with individual predicted peptides loaded on APCs. Measure activation via IFN-γ ELISA or upregulation of CD137.
- Engineered Variant Testing: Stimulate T-cell clones specific for a confirmed WT epitope with APCs loaded with the corresponding peptide sequence from the engineered Cas9 variant. Loss of activation confirms epitope disruption.
Visualizing Key Concepts and Workflows
Title: Rational Engineering Workflow for Low-Immunogenicity Cas9
Title: In Vitro T-Cell Immunogenicity Assay Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Cas9 Immunogenicity Research
| Item / Reagent | Function / Application | Example Vendor/Cat. No. (Illustrative) |
|---|---|---|
| Recombinant SpCas9 & Variants | Full-length antigen for in vitro and in vivo immunogenicity studies. Must be high-purity, low-endotoxin. | GenScript, Aldevron, Thermo Fisher. |
| HLA Tetramer (PE-conjugated) | Direct ex vivo staining and isolation of Cas9 epitope-specific T-cells. Custom-made for defined peptide-HLA complexes. | MBL International, Tetramer Shop. |
| Human IFN-γ ELISpot Kit | Quantification of antigen-specific T-cell responses at the single-cell level. | Mabtech, R&D Systems, BD Biosciences. |
| Pepsin or Lys-C | Enzymatic digestion of Cas9 protein to generate peptide fragments for epitope mapping studies. | Sigma-Aldrich, Promega. |
| netMHCIIpan Server | Computational tool for predicting peptide binding to HLA class II molecules, guiding epitope identification. | DTU Health Tech (online server). |
| Synthetic Peptide Pools | Overlapping 15-mer peptides spanning the Cas9 sequence for initial epitope screening. | JPT Peptide Technologies, GenScript. |
| Anti-Human CD137 (4-1BB) APC | Flow cytometry antibody to detect activated T-cells post-stimulation with Cas9 antigens. | BioLegend, BD Biosciences. |
| Cytokine ELISA Kits (IFN-γ, IL-2) | Quantify cytokine release from T-cell clones or lines in response to specific peptides. | Thermo Fisher, BioLegend. |
Benchmarking Immune Escape: Validating and Comparing Deimmunized Cas9 Platforms
The search for non-immunogenic CRISPR-Cas9 variants for therapeutic applications necessitates rigorous in vitro validation of predicted HLA class I and II epitopes. Computational prediction of immunodominant epitopes within the Cas9 protein must be empirically validated using gold-standard primary human immune cell assays. These systems assess the true potential of engineered Cas9 variants to trigger CD4+ and CD8+ T-cell responses, providing a critical bridge between in silico mapping and clinical safety.
Core Primary Human Immune Cell Assays
These assays utilize cells from healthy donors or specific HLA-typed cohorts to evaluate T-cell activation.
Peripheral Blood Mononuclear Cell (PBMC) Assay
A foundational assay to measure polyclonal T-cell responses.
Detailed Protocol:
- PBMC Isolation: Draw fresh human blood into heparin or EDTA tubes. Dilute blood 1:1 with PBS. Carefully layer over Ficoll-Paque PLUS density gradient medium. Centrifuge at 400-500 × g for 30-40 minutes at room temperature with brakes off. Collect the buffy coat layer at the interface. Wash cells twice with PBS and resuspend in complete RPMI-1640 medium (10% human AB serum, 1% penicillin-streptomycin, 1% L-glutamine).
- Antigen Presentation: Plate PBMCs (1-2 × 10^5 cells/well) in a 96-well U-bottom plate. Add test antigens:
- Peptide Pools: Overlapping 15-mer peptides (e.g., spanning the entire Cas9 sequence) at 1-2 µg/mL per peptide.
- Full-Length Protein: Recombinant wild-type or engineered Cas9 protein (1-10 µg/mL).
- Controls: Positive control (anti-CD3/CD28 beads, SEB), negative control (DMSO vehicle), and irrelevant peptide/protein.
- Incubation: Culture for 6-7 days at 37°C, 5% CO2.
- Readout (Day 7): Stimulate with the same antigen for an additional 24 hours, adding Brefeldin A for the final 4-6 hours. Perform intracellular cytokine staining (ICS) for IFN-γ, TNF-α, and IL-2, followed by flow cytometry analysis of CD4+ and CD8+ T-cells.
Dendritic Cell (DC) – T-Cell Co-culture System
This system models the natural antigen presentation pathway, crucial for evaluating cross-presentation of Cas9 epitopes.
Detailed Protocol:
- Monocyte-Derived DC (moDC) Generation: Isolate CD14+ monocytes from PBMCs using magnetic bead separation. Culture monocytes in complete RPMI-1640 medium supplemented with 100 ng/mL recombinant human GM-CSF and 50 ng/mL IL-4 for 5-6 days. Refresh cytokines on day 3.
- Antigen Loading & DC Maturation: On day 5-6, harvest immature DCs. Load with antigen: incubate with Cas9 protein (10-20 µg/mL) or peptide pools for 4-6 hours. Add maturation cocktail (e.g., TNF-α, IL-1β, IL-6, PGE2) and culture overnight.
- Autologous T-Cell Isolation: Isolate CD4+ or CD8+ T-cells from the same donor's PBMCs using negative selection kits.
- Co-culture: Plate matured, antigen-loaded DCs with autologous T-cells at a ratio of 1:10 to 1:20 (DC:T-cell) in a 96-well plate. Culture for 7-10 days.
- Readout: Re-stimulate T-cells with antigen-pulsed DCs or peptide-pulsed feeder cells. Measure T-cell proliferation (CFSE dilution) and cytokine production (ELISpot or multiplex cytokine assay).
Table 1: Quantitative Data Summary from Representative Cas9 Immunogenicity Studies
| Assay Type | Cell Source | Antigen Form | Key Readout | Observed Response (Wild-type Cas9) | Reference (Example) |
|---|---|---|---|---|---|
| PBMC Assay | HLA-diverse donors | Overlapping 15-mer peptide pools | IFN-γ ELISpot (SFU/10⁶ PBMCs) | 50-200 SFU (donor-dependent) | Wagner et al., 2019 |
| DC-T-cell Co-culture | HLA-A*02:01+ donors | Full-length S.pyogenes Cas9 protein | % Antigen-specific CD8+ T-cells (HLA-multimer+) | 0.1% - 0.8% of CD8+ T-cells | Ferdosi et al., 2021 |
| HLA-II Tetramer Assay | HLA-DR-typed donors | Predicted HLA-II binding peptides | Tetramer+ CD4+ T-cell frequency | Varies by peptide/HLA allele; up to 0.05% of CD4+ T-cells | Recent (2023) conference data |
| Cytokine Multiplex | PBMCs from pre-exposed donors | Cas9 protein | Secreted IFN-γ, IL-5, IL-13 (pg/mL) | IFN-γ: >500 pg/mL in responsive donors | Simhadri et al., 2022 |
Experimental Workflow for Cas9 Epitope Validation
The logical progression from prediction to validation involves sequential assays.
Diagram 1: Cas9 Epitope Validation Workflow
Key Signaling Pathways in T-Cell Activation by Cas9 Antigen
Understanding the molecular pathways triggered during assay readouts is essential.
Diagram 2: T-Cell Activation by Cas9-Derived Peptides
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for Primary Immune Cell Assays
| Reagent / Material | Function & Application in Cas9 Epitope Studies | Example Vendor/Product |
|---|---|---|
| Ficoll-Paque PLUS | Density gradient medium for isolation of viable PBMCs from whole blood. | Cytiva |
| Recombinant Human GM-CSF & IL-4 | Cytokines for differentiation of monocytes into immature dendritic cells (moDCs). | PeproTech |
| Human Leukocyte Serum (AB Serum) | Serum supplement for T-cell and DC media; reduces non-specific activation vs. FBS. | Valley Biomedical |
| HLA-Peptide Tetramers/Multimers | Fluorochrome-conjugated MHC-peptide complexes for direct staining of antigen-specific T-cells. | Immudex, MBL International |
| IFN-γ ELISpot Kit | High-sensitivity assay to quantify antigen-responsive T-cells via cytokine capture. | Mabtech |
| Intracellular Cytokine Staining (ICS) Antibody Panel | Antibodies for flow cytometric detection of cytokines (IFN-γ, TNF-α, IL-2) and surface markers (CD3, CD4, CD8). | BioLegend, BD Biosciences |
| Overlapping Peptide Libraries | Custom 15-mer peptides overlapping by 10-11 aa, spanning the entire Cas9 protein for epitope mapping. | JPT Peptide Technologies |
| Recombinant Cas9 Proteins | Wild-type and engineered, endotoxin-free Cas9 proteins for full-antigen presentation assays. | Applied StemCell, internal purification. |
| Magnetic Cell Separation Kits | For isolation of specific subsets (CD14+ monocytes, CD4+/CD8+ T-cells) with high purity. | Miltenyi Biotec, STEMCELL Technologies |
| Cellular Proliferation Dye (e.g., CFSE) | Fluorescent dye to track and quantify T-cell division over time in co-culture. | Thermo Fisher Scientific |
The advancement of CRISPR-Cas9-based therapeutics is critically dependent on understanding and mitigating host immune responses, particularly against the Cas9 nuclease. Research into Cas9 epitope mapping and the identification of immunodominant HLA epitopes requires sophisticated in vivo models capable of recapitulating the human immune system. This guide provides a technical comparison of the two foremost platforms for such immunological studies: humanized mouse models and non-human primate (NHP) studies. Each model offers distinct advantages and limitations for predicting T-cell and B-cell responses to Cas9 and other therapeutic proteins, directly impacting pre-clinical drug development.
Table 1: Core Characteristics & Capabilities
| Parameter | Humanized Mouse Models | Non-Human Primate (NHP) Studies |
|---|---|---|
| Human Immune System Fidelity | Partial; limited HLA diversity, often single allele; lacks full lymphoid organ architecture. | High; endogenous, fully functional immune system with complex MHC diversity and architecture. |
| Throughput & Scalability | High; allows for statistically powered N studies with multiple conditions. | Low; limited cohort sizes (typically 3-6), high cost per subject. |
| Experimental Timeline | Relatively short (12-20 weeks for engraftment + experiment). | Very long (years for chronic studies). |
| Capital & Operational Cost | Moderate (thousands USD per mouse). | Extremely high (hundreds of thousands USD per study). |
| Key Immune Cell Populations Present | Varies by model: HSC models yield myeloid & B cells; PBMC models yield primarily T cells. | Complete, endogenous repertoire of all immune cell types. |
| Applications in Cas9 Immunogenicity | Ideal for initial high-throughput screening of Cas9 variants for reduced HLA class I/II presentation. | Gold standard for final pre-clinical assessment of integrated immune response (innate & adaptive). |
Table 2: Data Output & Relevance to Epitope Mapping
| Data Type | Humanized Mouse Models | Non-Human Primate Studies |
|---|---|---|
| T-cell Epitope Mapping | Can identify dominant human T-cell responses in a restricted HLA context. Data may not represent full HLA diversity. | Identifies immunodominant epitopes in the context of a complete, polygenic MHC system. Responses are directly translatable. |
| Anti-Cas9 Antibody Titers | Can measure human IgG responses, but may lack germinal center optimization. | Provides comprehensive humoral response data, including affinity maturation and isotype switching. |
| Cytokine & Immunophenotyping | Flow cytometry on human cells from blood, spleen, and bone marrow. Limited tissue context. | Multiplex analysis from blood, lymph nodes, and tissues; provides systemic cytokine profiles. |
| Prediction Validation | Good for initial in vivo validation of in silico predicted epitopes. | Definitive validation of epitope immunodominance hierarchies and cross-reactivity. |
Detailed Experimental Protocols
Protocol 1: Cas9 Immunogenicity Assessment in Humanized NSG-SGM3 Mice
Objective: To evaluate human T-cell and antibody responses against wild-type and deimmunized Cas9 variants.
Materials: NOD.Cg-Prkdcscid Il2rgtm1Wjl Tg(CMV-IL3,CSF2,KITLG)1Eav/MloySzJ (NSG-SGM3) mice engrafted with human CD34+ hematopoietic stem cells (hu-NSG-SGM3).
Methodology:
- Engraftment & Validation: At 6-8 weeks, irradiate mice (1 Gy) and inject 1x10^5 human CD34+ cells via tail vein. Monitor engraftment weekly via flow cytometry for human CD45+ cells in peripheral blood. Proceed at >25% huCD45+ engraftment (typically 12-16 weeks post-transplant).
- Immunization: Formulate Cas9 protein (or mRNA/LNP) with a human-relevant adjuvant (e.g., AddaVax, a squalene-based emulsion mimicking MF59). Administer 20 µg dose via subcutaneous injection at week 0 and week 3.
- Sample Collection: Collect serum and peripheral blood mononuclear cells (PBMCs) at baseline, week 2, and week 5.
- Humoral Response Analysis: Measure anti-Cas9 IgG titers by ELISA using Cas9-coated plates and anti-human IgG-HRP.
- Cellular Response Analysis:
- Stimulate PBMCs with a library of overlapping 15-mer peptides spanning the Cas9 protein.
- Use IFN-γ ELISpot or intracellular cytokine staining (ICS) for IFN-γ, TNF-α, and IL-2 to identify reactive epitopes.
- For HLA restriction analysis, use antibodies to block specific HLA class I or II molecules during stimulation.
- Endpoint Analysis: Harvest spleen and bone marrow for deep immunophenotyping by spectral flow cytometry.
Protocol 2: Longitudinal Cas9 Immunogenicity Study in Cynomolgus Macaques
Objective: To comprehensively assess innate and adaptive immune responses to Cas9 delivery vectors (e.g., AAV, LNP) over time.
Materials: Naïve cynomolgus macaques (Macaca fascicularis), MHC-typed.
Methodology:
- Pre-study Screening: Select animals with diverse MHC haplotypes. Perform baseline immunology profiling: CBC, serum cytokines, PBMC immunophenotyping, and pre-existing anti-AAV/Cas9 antibody screening.
- Dosing: Administer Cas9 therapeutic (e.g., AAV-CRISPR construct) at a clinically relevant dose via the intended route (e.g., intravenous).
- Longitudinal Sampling: Collect blood at frequent intervals (e.g., D1, D3, D7, D14, M1, M3, M6). Schedule periodic lymph node fine-needle aspirates and bone marrow biopsies at key timepoints.
- Innate Immunity Profiling: Monitor acute phase reactants (CRP), complement activation, and myeloid cell activation via transcriptomics (NanoString) and cytokine multiplex panels (Luminex) on serum.
- Adaptive Immunity Deep Dive:
- T-cells: Perform high-parameter flow cytometry on PBMCs to track activation (CD38, HLA-DR), memory differentiation, and exhaustion (PD-1) markers on Cas9-specific T-cells identified by MHC multimer staining.
- B-cells: Use antigen-specific B-cell sorting with labeled Cas9 protein, followed by single-cell BCR sequencing to track clonal evolution and affinity maturation.
- Challenge/Re-dosing: At 6-12 months, administer a second dose of the vector to assess impacts of pre-existing immunity.
Visualizations
Diagram 1: Model Selection Pathway for Immunogenicity Testing
Diagram 2: Key Immune Pathways in Cas9 Response Across Models
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials forIn VivoImmunogenicity Studies
| Reagent/Material | Function & Application | Example/Catalog Consideration |
|---|---|---|
| Human CD34+ HSCs | Creation of humanized mouse models. Source (cord blood, mobilized peripheral blood) and purity critically impact immune reconstitution. | Freshly isolated or cryopreserved, >95% purity via CD34+ selection. |
| NSG & NSG-SGM3 Mice | Immunodeficient mouse strains that support engraftment of human immune cells. SGM3 variant expresses human cytokines for improved myeloid development. | The Jackson Laboratory: Stock # 005557 (NSG), # 013062 (NSG-SGM3). |
| Recombinant Cas9 Proteins/Peptides | Antigen source for immunization and ex vivo immune assays. Critical for epitope mapping studies. | Commercial suppliers (e.g., Aldevron, ACROBiosystems) or in-house purification with endotoxin control. |
| HLA/MHC Multimers (Tetramers, Dextramers) | Direct ex vivo staining and isolation of Cas9-specific T-cells from mouse or NHP samples. Custom reagents required for specific epitopes. | Custom synthesis services (e.g., Immudex, MBL International). |
| Adjuvants for Human Systems | To enhance immune responses in humanized mice, requiring adjuvants compatible with human cells. | AddaVax (MF59 mimic), CpG ODN (TLR9 agonist), Poly(I:C) (TLR3 agonist). |
| High-Parameter Flow Cytometry Panels | Deep immunophenotyping of human or NHP immune cells from limited samples. | Antibody panels for T-cell (activation, memory, exhaustion), B-cell, and myeloid lineages from BioLegend, BD Biosciences. |
| Single-Cell BCR/TCR Sequencing Kits | To analyze the clonal dynamics and specificity of antigen-specific B- and T-cell responses. | 10x Genomics Chromium Immune Profiling, Takara Bio SMART-Seq. |
| Cynomolgus Macaque MHC Typing Reagents | Essential for understanding genetic restrictions of immune responses in NHP studies. | Species-specific PCR-SSP or NGS-based typing services (e.g., Bio-Rad, GenDx custom solutions). |
This whitepaper provides an in-depth technical analysis of the immunogenic profiles of two primary CRISPR-Cas9 nucleases derived from Streptococcus pyogenes (SpCas9) and Staphylococcus aureus (SaCas9), in comparison to engineered, immune-evading orthologs. The context is a broader thesis on Cas9 epitope mapping and the identification of immunodominant HLA epitopes—a critical barrier to the clinical translation of CRISPR-based therapeutics. Preexisting humoral and cellular immunity against these bacterial-derived proteins can lead to reduced efficacy and potential adverse events, necessitating a detailed comparative understanding of their immunogenicity.
Core Immunogenicity Data
The following tables summarize quantitative data from recent studies on preexisting immunity and immune responses elicited by wild-type and engineered Cas9 orthologs.
Table 1: Preexisting Humoral Immunity in Human Populations
| Cas9 Variant | Seroprevalence (IgG) | Neutralizing Antibody Prevalence | Key Epitope Regions (from mapping studies) | Primary References |
|---|---|---|---|---|
| SpCas9 (Wild-type) | 58-78% | 15-40% | RuvC-III, HNH, PI domains; α-helical lobe | Charlesworth et al. (2019), Wagner et al. (2019) |
| SaCas9 (Wild-type) | ~24% | ~4% | WED-III, RuvC-II domains | Charlesworth et al. (2019) |
| Engineered eSpCas9 | Data correlated with SpCas9; epitope masking reduces neutralization. | <5% (in vitro assay) | Engineered surface residues (e.g., K848A, K1003A) reduce antibody binding. | Mehta & Merkel (2020) |
| Engineered SaCas9-KKH | Similar baseline to SaCas9; structural modifications may alter epitope presentation. | Not fully quantified | Modifications in REC3/WED domains potentially alter antigenicity. | Moreno et al. (2020) |
Table 2: T-cell Reactivity and HLA-Restricted Epitopes
| Cas9 Variant | IFN-γ ELISpot Frequency (SFU/10⁶ PBMCs) | Immunodominant CD4+ T-cell Epitopes (HLA-II Restricted) | Immunodominant CD8+ T-cell Epitopes (HLA-I Restricted) | Primary References |
|---|---|---|---|---|
| SpCas9 | 50-180 | DRB107:01, DRB115:01 peptides (e.g., aa 541-555, aa 991-1005) | A02:01, B07:02 peptides (e.g., aa 301-309, aa 766-774) | Ferdosi et al. (2019), Crudele et al. (2018) |
| SaCas9 | 10-45 | DRB101:01, DRB104:01 peptides (e.g., aa 201-215) | Less characterized; predicted lower frequency. | Simhadri et al. (2017) |
| Engineered iSpCas9 (LaserGene) | <20 | Epitope deletion/mutation in dominant HLA-II peptides (e.g., P991A, F1011S). | Mutation of HLA-I anchor residues in dominant peptides. | Mehta et al. (2022) |
Detailed Experimental Protocols
Protocol 1: Epitope Mapping via Peptide Scanning Arrays
- Objective: Identify linear B-cell and T-cell epitopes within Cas9 proteins.
- Methodology:
- Peptide Library Design: Synthesize a library of 15-20mer peptides spanning the entire Cas9 amino acid sequence, with 5-10aa overlaps.
- B-cell Epitope Mapping (ELISA): Coat ELISA plates with individual peptides. Incubate with human serum samples (diluted 1:100). Detect bound IgG using HRP-conjugated anti-human IgG and a colorimetric substrate. Signal >3x background indicates positive reactivity.
- T-cell Epitope Mapping (IFN-γ ELISpot): Isolate PBMCs from donors. Seed cells into ELISpot plates pre-coated with anti-IFN-γ antibody. Stimulate with individual peptides (10 µg/mL). After 24-48h, develop plates with biotinylated detection antibody, streptavidin-ALP, and BCIP/NBT substrate. Count spot-forming units (SFUs) using an automated reader.
- HLA Restriction Analysis: For positive peptides, co-incubate with anti-HLA-I or anti-HLA-II blocking antibodies or use antigen-presenting cells with defined HLA haplotypes to confirm restriction.
Protocol 2: In Vitro Neutralization Assay for Cas9 Activity
- Objective: Quantify the ability of serum antibodies to inhibit Cas9 ribonucleoprotein (RNP) function.
- Methodology:
- RNP Complex Formation: Pre-complex purified Cas9 protein (100 nM) with target-specific sgRNA (120 nM) for 10 min at 25°C.
- Serum Incubation: Incubate the RNP complex with serial dilutions of heat-inactivated human serum (1:10 to 1:1000) for 30 min at 37°C.
- Cell Transfection: Deliver the serum-treated RNP into reporter cell lines (e.g., HEK293 expressing GFP, with a Cas9 target site in GFP) via electroporation or lipofection.
- Activity Readout: After 72h, analyze GFP disruption via flow cytometry. Calculate % neutralization as:
[1 - (% editing with immune serum / % editing with naive serum)] * 100. Report the NT₅₀ (serum dilution for 50% neutralization).
Visualizations
Title: Immune Response Pathway Against Therapeutic Cas9
Title: Rational Design of Immune-Evading Cas9 Orthologs
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function / Application | Key Considerations |
|---|---|---|
| Recombinant Cas9 Proteins (Sp, Sa, Orthologs) | Direct antigen for ELISA, in vitro neutralization assays, T-cell stimulation. | Ensure >95% purity, endotoxin-free (<0.1 EU/µg) to avoid non-specific immune activation. |
| Overlapping Peptide Libraries | High-throughput mapping of linear B-cell and T-cell epitopes. | 15-20mer peptides with 5-10aa overlaps; purity >70%. Critical for ELISpot/Intracellular Cytokine Staining (ICS). |
| HLA-Typed PBMCs | Essential for defining HLA restriction of identified epitopes. | Use commercially available characterized panels or recruit typed donors. |
| IFN-γ ELISpot Kit | Gold-standard for quantifying antigen-specific T-cell responses. | High sensitivity; requires minimal cell numbers. Choose kits with pre-coated plates for consistency. |
| Reporter Cell Line with Integrated Target Site | Functional readout for Cas9 activity and neutralization assays. | Stable GFP or Luciferase reporters with an integrated, editable target sequence provide robust, quantitative editing data. |
| Anti-HLA Blocking Antibodies (Class I & II) | Confirm HLA restriction of T-cell epitopes. | Use in T-cell activation assays to inhibit response when the relevant HLA is blocked. |
| CRISP-Cas9 Knockout Cell Lines (e.g., B2M KO, CIITA KO) | Study antigen presentation pathways. | B2M KO lacks HLA-I; CIITA KO lacks HLA-II. Useful tools to dissect CD8+ vs. CD4+ T-cell responses. |
This technical guide assesses the critical functional trade-offs between nuclease activity, specificity, and delivery compatibility of CRISPR-Cas9 systems. This analysis is framed within a broader thesis focused on Cas9 epitope mapping and immunodominant HLA epitopes research. A primary translational challenge is that the human adaptive immune system frequently recognizes wild-type Streptococcus pyogenes Cas9 (SpCas9), generating neutralizing antibodies and cytotoxic T-cell responses directed against immunodominant epitopes. These immune responses can severely limit the efficacy and safety of in vivo therapeutic applications. Therefore, engineering Cas9 variants with reduced immunogenicity—through epitope masking, deimmunization, or alternative orthologs—must be rigorously evaluated against the core functional metrics of nuclease activity, on-target specificity, and compatibility with clinically relevant delivery vectors. This guide provides the experimental framework for this essential tripartite assessment.
Core Functional Metrics: Definitions and Quantitative Benchmarks
The performance of any Cas9 variant, including immunogenicity-optimized versions, is defined by three interdependent parameters.
Nuclease Activity: The efficiency of DNA cleavage at the intended target site, typically measured as indel frequency. High activity is non-negotiable for therapeutic efficacy. Specificity: The propensity to cleave at off-target genomic sites with sequence homology to the guide RNA (gRNA). High specificity is critical for safety. Delivery Compatibility: The physical packaging efficiency, stability, and functional delivery of the Cas9-gRNA complex via viral (e.g., AAV, lentivirus) or non-viral (e.g., LNPs, electroporation) vectors, constrained by cargo size and biocompatibility.
Trade-offs are inherent: enhancing specificity through high-fidelity mutations often reduces activity; deimmunizing mutations can destabilize the protein or alter its charge, affecting packaging; smaller Cas9 orthologs for AAV packaging may have different sequence preferences or reduced activity.
Experimental Protocols for Integrated Assessment
A robust evaluation requires parallel, standardized assays.
Protocol 3.1: Dual-Reporter Nuclease Activity & Specificity Assay
This method quantifies both on-target activity and off-target susceptibility in a single cellular system.
- Cell Line: Utilize HEK293T cells stably integrating two fluorescence reporter constructs.
- On-target Reporter: A constitutively expressed BFP gene disrupted by an in-frame STOP cassette flanked by the exact on-target cut site. Successful Cas9 cleavage and error-prone repair restores BFP fluorescence.
- Off-target Reporter: A constitutively expressed GFP gene disrupted by a STOP cassette flanked by a known, top-predicted off-target sequence for the gRNA.
- Transfection: Co-transfect cells with a plasmid expressing the Cas9 variant and the specific gRNA. Include a GFP-only expression plasmid for normalization.
- Flow Cytometry Analysis: At 72-96 hours post-transfection, analyze cells.
- Activity Metric: %BFP+ cells among transfected (GFP+) population.
- Specificity Metric: Ratio of (%BFP+ / %GFP+) from the off-target reporter. A lower ratio indicates higher specificity.
Protocol 3.2: GUIDE-seq for Genome-wide Specificity Profiling
For unbiased, genome-wide off-target detection.
- Oligonucleotide Transduction: Electroporate cells with the Cas9:gRNA RNP complex alongside a blunt, double-stranded GUIDE-seq oligonucleotide tag.
- Genomic DNA Harvest & Processing: Harvest genomic DNA after 72 hours. Shear DNA and prepare sequencing libraries, using primers that capture junctions between the genomic integration site of the tag and flanking DNA.
- Bioinformatic Analysis: Sequence libraries and use the GUIDE-seq analysis pipeline to identify genomic sites enriched with the tag sequence, indicative of double-strand breaks. Compare the number and location of off-target sites between wild-type and engineered/high-fidelity Cas9 variants.
Protocol 3.3: AAV Packaging andIn VitroPotency Assay
To assess delivery compatibility.
- Vector Production: Package the expression cassette for the Cas9 variant (driven by a compact promoter, e.g., EFS) and its gRNA (U6 promoter) into a recombinant AAV2 (capsid) vector via triple transfection in HEK293 cells. Purify via iodixanol gradient.
- Titration: Quantify vector genome (vg) titer via ddPCR using primers/probes against the Cas9 or gRNA scaffold.
- Functional Potency Assay: Transduce HEK293 cells harboring a stable GFP-to-BFP conversion reporter (e.g., at the EMX1 locus) with a range of AAV MOIs (e.g., 10^3 to 10^5 vg/cell). Measure the percentage of BFP+ cells via flow cytometry at 7-10 days post-transduction. Plot dose-response curve to determine functional titer (vg required for 50% conversion).
Table 1: Comparative Performance of Representative Cas9 Variants
| Cas9 Variant | Key Modification(s) | Avg. On-Target Indel % (HEK293, EMX1 site) | Relative Activity (vs. SpCas9) | GUIDE-seq Detected Off-Target Sites (Median) | Primary Rationale | AAV Packaging Compatible? (Size Limit: ~4.7kb) |
|---|---|---|---|---|---|---|
| SpCas9 (WT) | Reference Standard | 35-45% | 1.00 | 10-15 | Benchmark | No (cassette >4.7kb) |
| SpCas9-HF1 | Helical domain fidelity mutations (N497A/R661A/Q695A/Q926A) | 25-35% | ~0.65-0.75 | 1-2 | Enhanced Specificity | No |
| eSpCas9(1.1) | Positive charge reduction in non-target strand groove (K848A/K1003A/R1060A) | 30-40% | ~0.70-0.85 | 1-3 | Enhanced Specificity | No |
| SaCas9 | Staphylococcus aureus ortholog | 15-30% | ~0.40-0.60 | Often fewer than SpCas9 | Small Size for AAV | Yes (cassette ~4.2kb) |
| Cas9 Minimized | Immunodominant Epitope Deletion(s) | Variable (10-40%)* | Variable* | Variable* | Reduced Immunogenicity | Possibly (size reduced) |
*Data dependent on specific epitope modifications; must be empirically determined using Protocols 3.1 & 3.2.
Table 2: AAV-Delivered SaCas9 Potency Data (Example)
| Target Locus (in Reporter Cell Line) | AAV Serotype | MOI (vg/cell) | Transduction Efficiency (% GFP+ Cells) | Editing Efficiency (% BFP+ of Transduced) | Functional Titer (vg per 50% Editing) |
|---|---|---|---|---|---|
| EMX1 | AAV2 | 1 x 10^4 | ~95% | ~5% | 1.2 x 10^5 |
| EMX1 | AAV2 | 1 x 10^5 | ~98% | ~18% | 1.2 x 10^5 |
| VEGFA | AAV2 | 1 x 10^5 | ~98% | ~12% | 2.5 x 10^5 |
Visualization of Experimental Workflows and Relationships
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in Assessment | Key Consideration for Epitope Research |
|---|---|---|
| Dual-Fluorescence Reporter Cell Lines (e.g., HEK293-BFP/GFP-OT) | Provides integrated, flow-cytometry-based readout for on-target activity and a key off-target site in a single experiment. | Cell line must be selected for HLA haplotypes relevant to human immunogenicity studies (e.g., expressing HLA-A*02:01). |
| GUIDE-seq Oligonucleotide Tag | A short, double-stranded, end-protected DNA oligo that integrates into Cas9-induced DSBs for genome-wide off-target identification. | Unbiased detection is critical for assessing if deimmunizing mutations alter specificity profiles. |
| Recombinant AAV Serotype Kits (e.g., AAV2, AAV6, AAV-DJ) | Enables production of viral vectors for delivery compatibility and functional potency testing. | Capsid choice itself is immunogenic; consider alongside Cas9 immunogenicity. |
| HLA-Typed Primary Human Cells (e.g., PBMCs, T-cells) | The ultimate system for validating reduced immunogenicity of engineered Cas9 variants via T-cell activation assays (ELISpot, ICS). | Directly tests the hypothesis that epitope modification ablates immunodominant HLA-restricted responses. |
| Cas9-specific T-cell Reagents (Tetramers/Dextramers) | Peptide-MHC complexes used to detect and isolate T-cells specific for known immunodominant Cas9 epitopes. | Essential tool for epitope mapping and confirming the loss of reactivity against engineered variants. |
| High-Fidelity DNA Polymerase for GUIDE-seq NGS Lib Prep | Ensures accurate amplification of tagged genomic fragments prior to sequencing. | Critical for minimizing PCR-introduced noise in off-target detection. |
Within the broader thesis on Cas9 epitope mapping and the identification of immunodominant HLA-restricted epitopes, a critical translational step is the rigorous correlation of in vitro and ex vivo immunological data with potential clinical risks. This guide details the methodologies and frameworks required to integrate epitope mapping data into formal regulatory and safety assessments for therapeutic proteins, with a focus on CRISPR-Cas9 nucleases and other novel biologics.
Foundational Concepts: Epitopes, HLA, and Clinical Risk
Immunodominant Epitopes: Peptide sequences from a therapeutic protein that are efficiently processed, presented by Human Leukocyte Antigen (HLA) molecules, and elicit a robust T-cell response. Clinical Risk Correlates: Undesirable immune responses can lead to:
- Reduced therapeutic efficacy (neutralizing antibodies, cellular clearance).
- Acute infusion reactions (cytokine release).
- Off-target tissue damage (cross-reactive T-cells).
- Long-term loss of tolerance.
Quantitative Data: Epitope Frequency and Risk Indicators
Table 1: Correlating Epitope Characteristics with Clinical Risk Tiers
| Epitope Characteristic | Low Risk Indicator | Moderate Risk Indicator | High Risk Indicator | Measurement Assay |
|---|---|---|---|---|
| Population Frequency (HLA Restriction) | HLA allele frequency <1% in target population | HLA allele frequency 1-5% | HLA allele frequency >5% (e.g., HLA-A*02:01) | HLA genotyping databases (e.g., Allele Frequency Net Database) |
| In Vitro Immunogenicity (SI or % Responding) | Stimulation Index (SI) <2 or <5% donor response | SI 2-5 or 5-15% donor response | SI >5 or >15% donor response | T-cell ELISpot, Intracellular Cytokine Staining |
| Predicted Binding Affinity (IC50 nM) | IC50 >500 nM | IC50 50-500 nM | IC50 <50 nM (strong binder) | In silico prediction (NetMHCpan, IEDB tools) |
| Epitope Conservation (vs. Human Proteome) | No homology to human sequences | Low homology (≤5-mer match) | High homology (≥7-mer match) to human protein | BLASTp against human proteome |
| Pre-existing T-cell Memory (in naive donors) | Not detected in any donor | Detected in low frequency (<0.1% of CD4+/CD8+) | Detected in multiple donors (≥0.1% frequency) | MHC multimer staining, Antigen-specific T-cell expansion |
Table 2: Key HLA Alleles and Associated Risks for Cas9 Therapeutics
| HLA Allele | Global Approx. Frequency (%) | Known Risk for Immunogenicity | Common in Clinical Trials? | Risk Mitigation Strategy |
|---|---|---|---|---|
| HLA-DRB1*15:01 | ~10% (Caucasian) | High risk for antibody development to protein therapeutics | Yes | Deimmunization or exclusion in trial design |
| HLA-A*02:01 | ~25% (Global) | Dominant for CD8+ T-cell responses; high pre-existing memory risk | Ubiquitous | Critical epitope screening required |
| HLA-DRB1*07:01 | ~15% (Caucasian) | Associated with T-helper responses to biologics | Yes | Monitor in Phase I safety |
| HLA-B*07:02 | ~10% (Global) | Common presenter for viral & therapeutic peptides | Yes | Assess for cross-reactivity |
Experimental Protocols for Risk Correlation
Protocol: HLA-Donor Matched T-Cell Assay for Pre-Clinical Risk Assessment
Objective: To evaluate T-cell responses to predicted epitopes using donor PBMCs matched to the HLA-restriction of interest. Materials: See "Scientist's Toolkit" below. Method:
- Donor Selection: Isolate PBMCs from healthy donors (n≥50) characterized for HLA Class I and II alleles. Over-represent alleles of high frequency (e.g., HLA-A02:01, HLA-DRB115:01).
- Peptide Pools: Synthesize 15-mer peptides overlapping by 11 amino acids spanning the entire therapeutic protein. Also synthesize individual predicted immunodominant epitopes (9-12mers).
- T-Cell Expansion (Day 0-10): Culture 1x10^6 PBMCs/well with peptide pools (1 µg/mL/peptide) or individual epitopes in RPMI-1640 + 10% human AB serum + 50 IU/mL IL-2. Include positive (anti-CD3/CD28) and negative (DMSO) controls.
- ELISpot Assay (Day 10-12): Harvest cells. Seed 1x10^5 cells/well on IFN-γ pre-coated ELISpot plates. Re-stimulate with corresponding peptides (5 µg/mL) for 24h. Develop plates per manufacturer's protocol.
- Data Analysis: Count spot-forming units (SFU). A positive response is defined as SI ≥2 (SFU peptide / SFU negative control) and ≥50 SFU/10^6 cells above background. Calculate responder frequency (% donors positive for each epitope).
Protocol: MHC Multimer Staining for Pre-existing Memory
Objective: To detect pre-existing CD8+ T-cell memory against Cas9-derived epitopes in naive donors. Method:
- Multimer Generation: Generate PE- or APC-conjugated MHC Class I dextramers (e.g., HLA-A*02:01) loaded with predicted strong-binding Cas9 epitopes.
- Staining: Incubate 1x10^6 fresh, unstimulated PBMCs from HLA-matched donors with multimer (10 µL) for 15 min at 4°C in the dark. Add surface antibodies (anti-CD3, CD8, CD19, CD14).
- Exclusion & Analysis: Exclude CD19+ B cells and CD14+ monocytes. Analyze by flow cytometry. Pre-existing memory is defined as multimer+ CD8+ T-cells ≥0.1% of total CD8+ population in the absence of in vitro expansion.
Visualizing the Risk Assessment Workflow & Immunological Pathways
Title: Clinical Immunogenicity Risk Assessment Workflow
Title: T-Cell Activation Pathway Leading to Clinical Risk
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Epitope-Clinical Risk Correlation Studies
| Item | Function/Application | Example Vendor/Product |
|---|---|---|
| HLA-Typed PBMCs | Provide biologically relevant, genetically diverse immune cells for in vitro assays. Essential for correlating response with specific HLA alleles. | Commercial biorepositories (e.g., StemCell Technologies, AllCells). |
| MHC-Peptide Dextramers/Multimers | Directly stain and quantify antigen-specific T-cells (including pre-existing memory) from patient samples without in vitro expansion. | Immudex, ProImmune, MBL International. |
| IFN-γ/IL-5 ELISpot Kits | Gold-standard for quantifying T-cell responses (Th1/Th2) to specific epitopes. High sensitivity for low-frequency responses. | Mabtech, R&D Systems, ImmunoSpot. |
| Predicted Epitope Peptide Libraries | Custom synthetic peptide pools (overlapping or predicted) covering the entire protein for unbiased screening. | GenScript, PEPscreen libraries (Sigma), Aalto Bio. |
| Intracellular Cytokine Staining (ICS) Antibody Panels | Multiplex flow cytometry to characterize polyfunctional T-cell responses (IFN-γ, TNF-α, IL-2, IL-4, etc.) at single-cell level. | BioLegend, BD Biosciences Cytometric Bead Array. |
| In Silico Prediction Platforms | Prioritize epitopes for experimental validation using neural networks trained on binding and elution data. | IEDB Analysis Resource, NetMHCpan, NetMHCIIpan. |
| Cell Deconvolution Software | Analyze complex ELISpot or flow data, calculate responder frequencies, and perform statistical correlation with HLA alleles. | ImmunoSpot Analyzer, FlowJo, R/Bioconductor packages. |
The final correlation involves creating an Integrated Summary of Immunogenicity (ISI) for regulatory filings (e.g., EMA, FDA). This document must juxtapose in silico predictions, in vitro responder frequencies, HLA-risk correlations, and any in vivo data (if available from animal models). A clear argument must be presented on how identified high-risk epitopes have been mitigated through protein engineering or will be managed through clinical monitoring, patient screening, or dosing strategies. This systematic, data-driven correlation transforms epitope mapping from a research exercise into a cornerstone of modern biologic safety assessment.
Conclusion
The systematic mapping of Cas9 epitopes and their associated immunodominant HLA restrictions is no longer a niche pursuit but a fundamental pillar of responsible therapeutic development. This synthesis of foundational knowledge, methodological rigor, troubleshooting insights, and comparative validation provides a roadmap for mitigating the immunogenic risks of CRISPR-based therapies. The key takeaway is that successful deimmunization requires an integrated approach, combining sophisticated in silico prediction with robust experimental validation across diverse HLA backgrounds, all while vigilantly preserving nuclease function. Future directions point toward personalized epitope screening for patient stratification, the development of HLA-supertype-targeted Cas9 variants, and the creation of universal 'stealth' editors. Ultimately, a deep understanding of this immune landscape is the critical bridge that will carry CRISPR technology from potent laboratory tool to safe and effective mainstream medicine, unlocking its full therapeutic potential for diverse global populations.